Papaya black seeds have beneficial anticancer effects on PC-3 prostate cancer cells
Abstract
Aim: The study investigated the effect of papaya seeds on prostate cancer (PC) using PC-3 cell line because papaya seeds have effects on the male reproductive system notably decreasing sperm concentration, motility, and viability, leading to azoospermia after short-to-long-term treatment.
Methods: The black seeds from yellow (ripe) papaya and white seeds from green (unripe) papaya were harvested and then extracted in water, 80% methanol, and hexane. The cytotoxic effects of seeds extracts were determined using a WST-1 proliferation assay. The amount of total polyphenols was determined using Folin Ciocalteu reagent.
Results: The methanol extracts from black seeds significantly (P < 0.05) decreased cell proliferation of PC-3 cells whereas hexane- and water-extracts have no effect. However, the water-extract from white seeds stimulated PC cell proliferation. The black seeds contained significantly more polyphenols than that of white seeds. The data suggest that black seeds from papaya have anticancer effects on PCs whereas white seeds stimulated prostate cancer proliferation. The anticancer effect of black seeds may be because of their high concentration of polyphenols.
Conclusion: The black seeds from papaya may have a potential to reduce growth of prostate cells; however, consumption of white seeds should be avoided as they may stimulate pre-existing prostate cancer.
Keywords
Introduction
The prostate cancer is the malignant tumor of the prostate gland of male reproductive organ, which may be life threatening, when spread to other body parts, predominantly towards lymph nodes, and bones.[1] According to a report (2014) published by Cancer Research, UK, an estimated 14 million cases of cancer reported worldwide and nearly half-8.2 million people (about 13% of the total worldwide deaths) died from cancer.[2] The cases of cancer is expected to increase to 24 million by 2035, therefore, the death toll from cancer is expected to increase in the future.
The major factor contributing in prostate cancer is the age, because men who have the prostate cancer are between 65 and 80 years, and it is rare in men who are under than 40 years.[3] The other contributing factor is genetics. For example, men of African-American descent are at a significantly higher risk of developing prostate cancer than white men. In fact, prostate cancer is the fourth most common reason overall for death in African-American men. About 19% of black men, (1 in 5) will be diagnosed with prostate cancer, and 5% of those will die from this disease.[4] Also, a man who has a member of his family with prostate cancer is more likely to get the prostate cancer compared to another man.[5] In addition, a man who inherited the faulty BRCA2 gene is more likely to get the more severe type of prostate cancer.[5]
Obesity also contributes to prostate cancer.[6] Sex hormones are also involved in prostate cancer development. Reduced testosterone levels have been related to obesity, metabolic syndrome (MS), benign prostatic hypertrophy and even prostate cancer.[7] Diet rich in high-fat milk and red meat, saturated fats and omega-6 fatty acids was found to increase the risk of prostate cancer whereas diet rich in fruits and vegetable decreases the risk of prostate cancer.[8-10] An inverse association between high intake of vegetables and/or fruits and incidences of cancer was reported by a number of epidemiological studies.[11-17] Further, the preventive effect on prostate cancer risk was found for a diet, which was rich in tomato products and lycopene.[18] Unfortunately, there are conflicting findings on the lycopene-prostate cancer risk relationship and the preventive role of tomato products.[19,20]
Papaya fruits also contained a significant amount of lycopene and anticancer activities of papaya have been demonstrated in a number of in vitro studies.[21] Papaya juice and pure lycopene caused cell death in the liver cancer cell line, Hep G2.[22] Papaya seed extract exhibited anticancer activity in acute promyelotic leukemia HL-60 cells whereas papaya pulp extract did not have any effect.[23]
Papaya has also been used as a traditional medicine in some cultures for male fertility, suggesting its direct role in male reproductive system.[24] Papaya seeds are natural contraceptive for both man and women. It is traditionally used to affect the fertility in men in a reversible manner. Interestingly papaya seeds have not known for side effects, as these are common with pharmaceutical contraceptive.[24,25] It appears that Papaya seeds have activity for male reproductive system; it is, therefore, possible that papaya seeds may have anticancer effect against cancer of prostate gland, a vital organ of male reproductive system. We hypothesized that papaya seeds can be effective in inhibiting prostate cancer cells proliferation and, therefore, may be a good nutraceutical for preventing and/or treating prostate cancer in men. To determine the effect of papaya seeds extract on prostate cancer cell proliferation, we used both white and black seeds extracts for their anticancer activity using PC-3 prostate cancer cells.
Methods
Materials
PC-3 cells (CRL-1435) and 3T3L1 cells (CL-173) were purchased from ATCC (Manassas, VA 20110), F-12K and DMEM media was purchased from Gibco (Grand island, NY14072), Fetal Bovine Serum (FBS-BBT) was purchased from RAMBIO (Missoula, Montana), antibiotics penicillin and streptomycin (BP2959) and Phosphate Buffered Saline (PBS; BP399-550) was purchased from Fisher (Fair lawn, New Jersey 07410). Green papaya was obtained from Randolph Farm at Virginia State University, whereas golden papaya was purchased by Tex State Distributing LLC (Alaneo, TX). WST-1 (MK400) was purchased from Talkara (Shiga, Japan).
Preparation of papaya seeds extracts
The papaya was washed with distilled water, blotted dry with paper towel, and was cut into half to access the seeds. The seeds were scrapped and washed 3 times with distilled water. The washed seeds were spread on a plastic trays and left for drying in a chemical hood until a constant weight was obtained. The dried seeds were grounded to a fine powder using a mortar and pestle with liquid nitrogen added to keep the powder frozen. The dried powder was flashed with nitrogen and stored at -80 ℃ until used. A known quantity (5 g) of dried papaya powder was mixed with 100 mL of distilled water, 80% methanol, or hexane and placed on a shaker at room temperature overnight.
The next day, the mixture was centrifuged at 1,500 g for 20 min using a Thermo Scientific centrifuge (Waltham, MA). The supernatant was collected and the residues washed 2 times by suspending them again in the respective solutions, mixing, and placing on shaker overnight. The collected supernatant was pooled together and the residues were discarded. The hexane and methanol extracts were dried in a nitrogen evaporator (Organomation Associates, Inc, Berlin, MA) to dryness and then subjected to freeze drying over night to ensure removal of traces of solvents. The water extract was freeze dried. The dried extract was stored in a -20 ℃ freezer.
Cell culture
The PC-3 prostate cancer cells were cultured in F-12K media whereas 3T3L1 cells were cultured in DMBM media. Both media were supplemented with 10% FBS and 1% penicillin and Streptomycin. The cells were incubated in a humidified incubator at 37 ℃ with 5% CO2. Media was changed every 3 days and cells were subcultured when they became confluent.
Cell proliferation assay
Effect of papaya seeds extract on cell proliferation was determined using a WST-1 assay as per manufacturer instructions. The assay is based on the reduction of WST-1 dye (brown color) by mitochondrial dehydrogenases in viable cells. The reduced dye changes to an orange color and the intensity of color is proportional to number of living cells, which can be estimated by reading at 420 nm in a spectrophotometer. Cells (10,000/well) were initially incubated for 24 h in a 96 well plate as described above. For treatment with extracts, media was replaced by serum-free media containing varying amounts of papaya seeds extract. The dried water extract was dissolved in serum-free F-12 or DMEM media whereas dried methanol and hexane extracts were initially suspended in 50% DMSO. The concentration of stock solution was 250 mg/mL. The extracts were diluted with serum-free F-12 or DMEM media to make 0-250 µg/mL concentrations for treatment. The concentration of DMSO did not exceed to 0.05% and has no effect on cell viability.
Determination of total polyphenols
The extracts of papaya seed was used to determine the total polyphenols as described previously.[26] Briefly, the extracts was incubated with Folin Ciocalteu reagent (Sigma Chem. Co., St. Louis, MO) and the formation of a blue chromophore from the reduction of phosphotungstic phosphomolybdenum was determined at 765 nm. The total phenolic content was calculated from a calibration curve using Gallic acid as a standard, and the result are expressed as mg Gallic acid equivalent per g dry weight of sample.
Data analysis
The data is expressed as mean ± SD for at least 3 replicates. All comparisons were made by one-way ANOVA with Tukey’s-HSD-post-hoc test using SPSS Statistics 20 software. All significant differences are reported at P < 0.05 and indicated by “*”.
Results
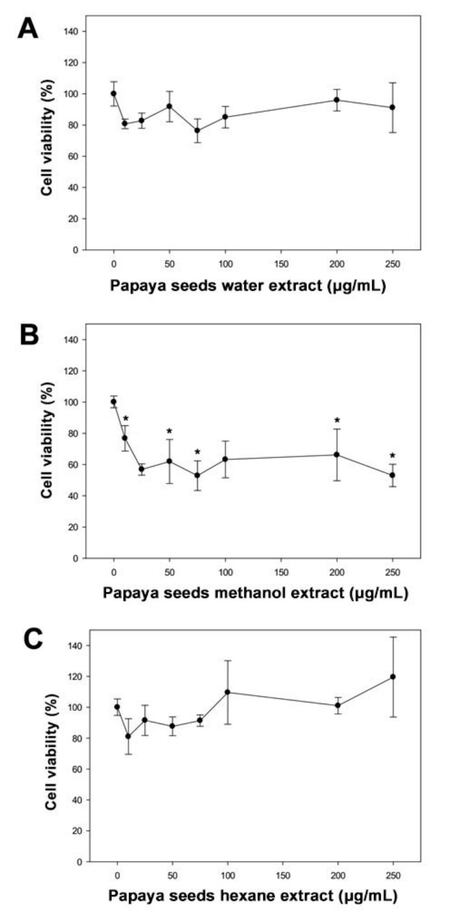
When cells were treated with water extract of papaya black seeds, the cells viability initially decreased slightly by 20% (non-significant) at 25 g/mL. The cells viability did not change further on increasing the papaya seeds extract [Figure 1A]. When cells were treated with methanol extract of papaya black seeds, the cells viability initially decreased significantly to 60% (P < 0.05) in a concentration depends manner up to 25 µg/mL; however, the cells viability was not decreased further by increasing the concentration of methanol extract over 25 µg/mL [Figure 1B]. However, when cells were treated with hexane extract of papaya black seeds, the cells viability didn’t show any significant change [Figure 1C].
Figure 1. The effect of black papaya seeds on PC-3 prostate cancer cells. Cells (10,000/well) were incubated with different concentration of water (A), methanol (B), and hexane (C) extracts of papaya black seeds in a CO2 incubator at 37 ℃ for 24 h. After treatment, cell viability was determined using a WST-1 assay. Results are expressed as mean ± SD for at least 3 replicates. All comparisons were made by one-way ANOVA with Tukey's-HSD-post-hoc test using SPSS Statistics 20 software. *Significant differences between treated and untreated groups were reported as *P < 0.05
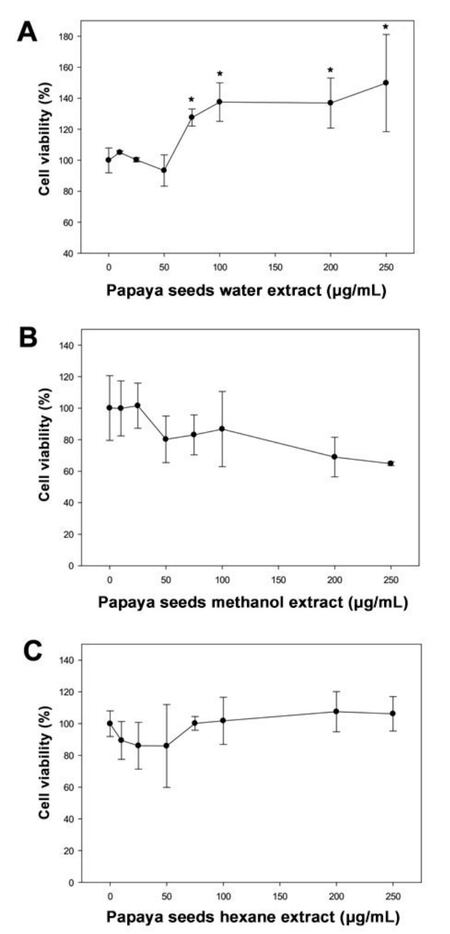
When cells were treated with water extract of papaya white seeds, the cells viability initially did not change up to 50 µg/mL; however, by further increasing the concentration of papaya extract, the cells proliferation increased significantly (P < 0.05) up to 140% [Figure 2A] at 250 g/mL. When cells were treated with methanol extract or hexane extracts of papaya white seeds, the cells viability initially decreased slightly by 20% or 10% (non-significant), respectively at 50 µg/mL. The cells viability did not change further on increasing the concentration of papaya seeds extract [Figure 2B and C].
Figure 2. The effect of white papaya seeds on PC-3 prostate cancer cells. Cells (10,000/well) were incubated with different concentration of water (A), methanol (B), and hexane (C) extracts of papaya white seeds in a CO2 incubator at 37 ℃ for 24 h. After treatment, cell viability was determined using a WST-1 assay. Results are expressed as mean ± SD for at least 3 replicates. *Significant differences between treated and untreated groups were reported as *P < 0.05
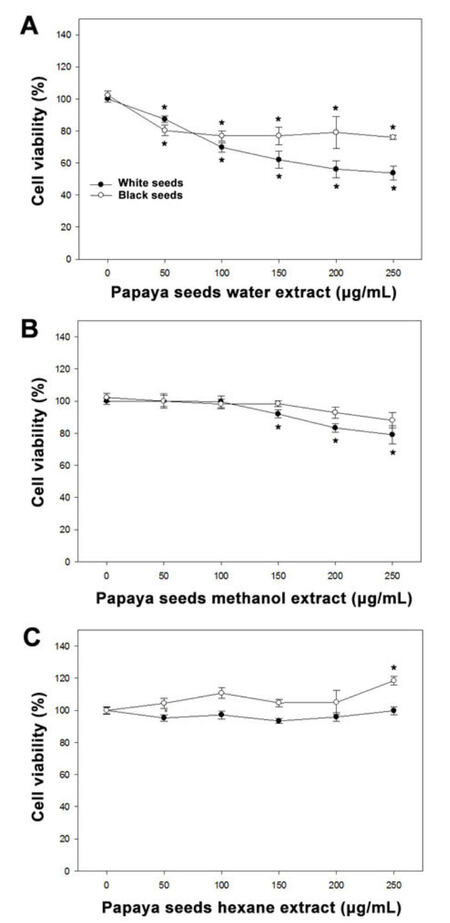
We have further tested methanol, hexane, and water extracts of black and white seeds on non-cancerous undifferentiated 3T3L1 fibroblasts [Figure 3]. The hexane extracts of either black or white seeds almost have no cytotoxic effect on the growth of 3T3L1 fibroblasts with the exception of black seeds extract that exhibited a marginal increase in cell proliferation (P < 0.05) at 250 µg/mL. The methanol extract of white seeds was also slightly cytotoxic (10-15%, P < 0.05) at 150 µg/mL or higher doses. In contrast, water extracts from both white and black seeds caused cytotoxic effects in these cells in a dose dependent manner. The white seed inhibited cell proliferation by 50% at 250 µg/mL whereas black seeds have a lesser effect and inhibited cell proliferation only by 25% at this concentration.
Figure 3. The effect of Papaya seeds extract on non-cancerous undifferentiated 3T3L1 fibroblasts. Cells (10,000/well) were incubated with different concentration of water (A), methanol (B), and hexane (C) extracts of papaya white or black seeds in a CO2 incubator at 37 ℃ for 24 h. After treatment, cell viability was determined using a WST-1 assay. Results are expressed as mean ± SD for at least 3 replicates. *Significant differences between treated and untreated groups were reported as *P < 0.05
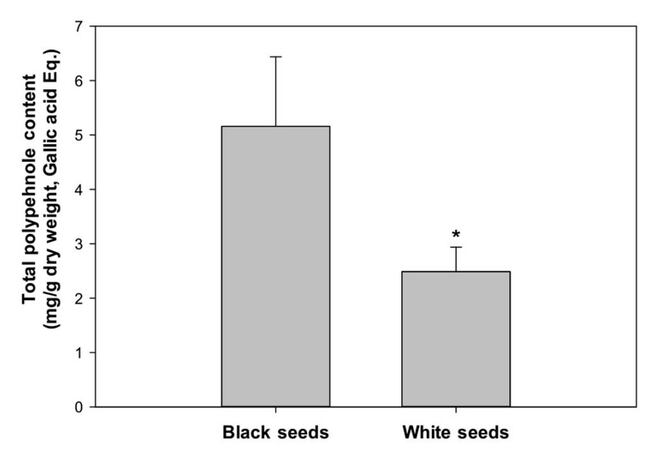
Data presented in Figure 4 indicates that black seeds contained significantly more polyphenols than that of white seeds. The total polyphenols were almost twice in black seeds than that in the white seeds (black seeds, 5.2 mg/g dry powder vs. white seeds 2.5 mg/g dry powder; P < 0.05).
Figure 4. Total phenolic contents in black and white papaya seeds. The phenolic contents in black and white extracts were determined using a Folin Ciocalteu reagent (Sigma Chem. Co., St. Louis, MO) and the total phenolic content was calculated from a calibration curve using Gallic acid as a standard, and the result are expressed as mg Gallic acid equivalent per g dry weight of sample. *Significant differences between black seeds and white seeds were reported as *P < 0.05
Discussion
The present study was conducted to examine the effects of papaya seeds extracts on prostate cancer. The seeds account for only 7% of papaya weight but typically discarded. Different parts of papaya have been used to prevent or treat a number of diseases. Oil extract of papaya seed have shown antifungal activity.[27] The fatty acids identified in the seed extracts of papaya (from ripe fruit) were able to reduce the number of Trypanosoma cruzi parasites from both parasite stages, blood trypomastigote and amastigote (intracellular stage) in mice.[28] The chloroform extract of the papaya seeds showed contraceptive efficacy without adverse toxicity, mediated through inhibition of sperm motility.[29] The methanolic extract of the seeds also showed antifungal activity against Aspergillus flavus, Candida albicans and Penicillium citrinium.[30] Furthermore, treatment of methanolic extract of the papaya seeds in rats induced gastroprotection without signs of toxicity. This effect seems to involve sulfhydryl compounds, increased mucus, and reduced gastric acidity.[31]
In addition to various biological activities described above, anticancer activities have been demonstrated by papaya extracts in a number of in vitro studies.[21] Papaya juice and pure lycopene, a component present in papaya, caused cell death in the liver cancer cell line, Hep G2, with the half maximal inhibitory concentration (IC 50) of 20 µg/mL and 22.8 µg/mL, respectively.[22] Papaya seed extract also exhibited anticancer activity in acute promyelotic leukemia HL-60 cells at IC 50 of 20 µg/mL whereas papaya pulp extract did not have any effect even at a concentration of 100 µg/mL.[23] In contrast, Garcia-Solis et al.[32] observed that papaya pulp inhibited of proliferation of MCF-7 cells after 72-h treatment. In our studies, we have observed almost 50% growth inhibition at 25 µg/mL of methanol seeds extract on PC-3 cells. These results are in the similar range of concentration as shown by other studies.[22,23] This effect does not appear to be due to residual methanol contamination because the methanolic seeds extract after dryness under nitrogen gas was subjected to freeze drying under vacuum over night to ensure complete removal of methanol. Furthermore, from both seeds extracts only methanolic white seeds extract has a small (15% inhibition) effect (P < 0.05) on 3T3L1 fibroblasts [Figure 3], again suggesting unlikely effect of methanol in the extracts.
The effect of papaya on cancer growth inhibition are further evident from studies performed by Morimoto et al.[33] who patented the extracts of different parts of papaya for the prevention, treatment, or improvement of many types of cancer, including stomach, lung, pancreatic, colon, liver, ovarian, neuroblastoma, lymphoma, leukemia, and other blood cancers. Effect of papaya leaves have also been characterized on T-cell lines, Burkitt’s lymphoma cell lines, chronic myelogenous leukemia cell line, cervical carcinoma cell line, hepatocellular carcinoma cell lines, lung adenocarcinoma cell line, pancreatic epithelioid carcinoma cell line 1, pancreatic adenocarcinoma cell line, mesothelioma cell lines, plasma cell leukemia cell line, anaplastic large cell lymphoma cell line, breast adenocarcinoma cell line (MCF-7), and mesothelioma cell line.[34] Other studies have shown effect of the aqueous extract of papaya seeds on human kidney epithelial cells, human colon epithelial cells, human lung fibroblasts, and human pancreatic cells.[35] Interestingly no human clinical trials were performed as yet; however, survival was observed in patients with lung cancer, stomach cancer, breast cancer, pancreatic cancer, liver cancer, and blood cancer after drinking papaya leaf extract.[21] One report suggested that papaya can be effective against prostate cancer because of its lycopene content;[36] however, to our knowledge no study has been performed to test the effect of papaya on prostate cancer. Furthermore, water extracts from both white and black seeds exhibited cytotoxic effects on non-cancerous undifferentiated 3T3 fibroblasts whereas only methanolic white seeds extracts have a marginal effect and hexane seeds extracts has no cytotoxic effects. The cytotoxic effect (15%) in the methanolic extracts from white seeds occurred at 250 µg/mL concentration which is about 10 times higher than a 50% inhibition which was observed only 25 µg/mL of methanolic black seeds extract. Typically all cancer drugs exhibit some degree of cytotoxic effects on normal cells. Although deviated from normal path, many pathways for cellular replication and repair are similar between cancer and normal cells. However, as cancer cells divide more rapidly than normal cells, the cytotoxic drugs more selectively kill cancer cells whereas the normal cells are able to adapt and recover from toxicity.[37]
We hypothesized that papaya seeds may possess anticancer activity on prostate cancer because of its effects on male reproductive system. The aqueous extracts from papaya seed have been reported to reversibly decrease the testicular weight and to suppress spermatogenesis, and fertility in rats.[38] This study suggested that water extract of papaya seeds suppresses the activities of steroidogenic enzymes in the testis of rats, and that this may contribute to reversible suppression of spermatogenesis, a property that gives a possible male contraceptive potential.[38] Other studies have shown that the seed extract resulted in a significant dose dependent suppression of cauda epididymal sperm motility coinciding with a decrease in sperm count and viability with no developmental toxicity and teratogenicity which could affect pregnancy, implantation, and gestation.[39] However, another study reported that low dose aqueous crude extract of papaya seeds did not adversely affect prenatal development, whereas high doses of papaya extracts resulted in abortifacient property indicating that papaya toxicity can adversely affect the fetus.[40] These studies suggest that papaya has wide range of activities on cellular targets in male reproductive system. We, therefore, tested the effect of papaya seeds on prostate cancer cells.
Unripe papaya contains white seeds whereas ripe papaya contains black seeds. The different color of seeds indicates that as papaya matures its compositions changes. The presence and absence of different compounds in papaya seeds, therefore, may have different biological properties. We used hexane, methanol, and water to extract compounds of different chemical nature. The hexane was used to extract non-polar compounds. During this study we did not see any effect of hexane extract from either black or white seeds on prostate cancer cells. However, it has been shown that hexane extract of the papaya seed was highly effective in inducing apoptosis or cell death in leukemic HL-60 cells.[23] Papaya seeds contain contained considerable amount of oil (27.0%),[41] comprising 45.9% of oleic acid, 24.1% of palmitic and 8.52% of stearic acid.[28] Among phytochemicals, the major constituent is benzyl isothiocyanate (99.36%).[27] The apoptotic effect of papaya seeds on HL-60 cells were comparable to those of authentic benzyl isothiocyanate.[27] However, lack of hexane effect on prostate cancer cells suggest that benzyl isothiocyanate may not has effect in these cells.
Our data indicate that methanol extract from black seeds was effective in inhibiting cell proliferation of prostate cancer cells whereas the methanol extract from white seeds extract was not effective. Alcoholic extracts are commonly used to extract phytochemicals as it solubilize most of the polyphenols including flavonoids and alkaloids.[42,43] The Phenolic compounds are a main class of secondary metabolites in plants and possess a number of potent biological activities including anti-oxidation, anti-cancer, anti-bacterial, and immune modulating activities.[44] We found that black seed extract have almost 2.5 times more polyphenolic contents then white seeds. It is, however, not known what compound is predominantly present in black versus white seeds. It is, therefore, difficult to predict a tentative active compound in our methanolic extract that resulted in prostate cancer cell growth inhibition. Further experiments are required to perform a comparative analysis of phytochemicals present in the black and white seeds to identify and test the potential anticancer agent in black seeds of papaya.
In contrast to methanolic seeds extract from papaya back seeds, we found that the aqueous extract of papaya white seeds, surprisingly, stimulated prostate cancer cell proliferation whereas aqueous extract of black seeds has no effect. Papaya seeds are rich in various types of phytochemicals including saponins, tannins, polyphenols, flavonols, glucosides, alkaloids, triterpenes, amino acids, sugars, proteins, and vitamins.[45] Some of the known phytochemicals identified by LCMS-MS technique in the aqueous extract of papaya seeds include 5-hydroxy feruloyl quinic acid, acetyl p-coumaryl quinic acid, quercetin-3-O-rhamnoside, syringic acid hexoside, 5-hydroxy caffeic quinic acid, peonidin-3-Oglucoside, sinapic acid-O-hexoside, cyaniding-3-O-glucose and methyl feruloyl glycoside.[46] Again, it is not clear if these phytochemicals are present in black seeds or white seeds. It is apparent from our study that some tumor promoting substance is present in white seeds which is no longer present as the white seeds mature into black seeds when papaya becomes ripe. Clearly, further studies are required to resolve this issue.
To our knowledge we performed first study to demonstrate the effect of papaya seeds on prostate cancer cells. Additional studies are required using other prostate cancer cell lines and cancer cells lines of different origin to validate the specificity of papaya black seeds extracts. The future studies will also be required to characterize phytochemical profile of papaya seeds, to identify targets of intracellular signaling pathways, to determine pharmacokinetics of the active compounds, and toxicological safety of the bioactive fraction in an in vivo animal model. These studies will be essential to pave the way for a successful clinical translation.
Declarations
Authors’ contributionsPerformed the experiments: K.S. Alotaibi
Analyzed the data: H. Li
Cultivated papaya and contributed in the experimental design: R. Rafi
Conceived and designed the study, and prepared the manuscript: R.A. Siddiqui
Financial support and sponsorshipThe work was supported by funds provided by Agriculture Research Station, Virginia State University, Petersburg, Virginia, USA.
Conflicts of interestThere are no conflicts of interest.
Patient consentNot applicable.
Ethics approvalNot applicable.
REFERENCES
1. Ruddon RW. Cancer Biology. 4th ed. London: Oxford University Press; 2007.
2. Cancer Statistics for UK (2014). Available from: http://www.cancerresearchuk.org/health-professional/cancer-statistics. [Last accessed on August 14, 2017].
4. Jazayeri SB, Samadi DB. Prostate cancer in African Americans: early oncological and functional outcomes after robotic prostatectomy. Int J Urol 2017;24:236-7.
6. Wu VJ, Pang D, Tang WW, Zhang X, Li L, You Z. Obesity, age, ethnicity, and clinical features of prostate cancer patients. Am J Clin Exp Urol 2017;5:1-9.
7. van der Kwast TH, Têtu B. Androgen receptors in untreated and treated prostatic intraepithelial neoplasia. Eur Urol 1996;30:265-8.
8. Masko EM, Allott EH, Freedland SJ. The relationship between nutrition and prostate cancer, is more always better? Eur Urol 2013;63:810-20.
9. Fabre B, Grosman H, Gonzalez D, Machulsky NF, Repetto EM, Mesch V, Lopez MA, Mazza O, Berg G. Prostate cancer, high cortisol levels and complex hormonal interaction. Asian Pac J Cancer Prev 2016;17:3167-71.
10. Williams G. Aromatase up-regulation, insulin and raised intracellular oestrogens in men, induce adiposity, metabolic syndrome and prostate disease, via aberrant ER-α and GPER signaling. Mol Cell Endocrinol 2012;351:269-78.
11. Lin PH, Aronson W, Freedland SJ. Nutrition, dietary interventions and prostate cancer: the latest evidence. BMC Med 2015;13:3.
12. Liu B, Mao Q, Cao M, Xie L. Cruciferous vegetables intake and risk of prostate cancer: a meta-analysis. Int J Urol 2012;19:134-41.
13. Ma RW, Chapman K. A systematic review of the effect of diet in prostate cancer prevention and treatment. J Hum Nutr Diet 2009;22:187-99; quiz 200-2.
14. Kenfield SA, Batista JL, Jahn JL, Downer MK, Van Blarigan EL, Sesso HD, Giovannucci EL, Stampfer MJ, Chan JM. Development and application of a lifestyle score for prevention of lethal prostate cancer. J Natl Cancer Inst 2015;108: doi: 10.1093/jnci/djv329.
15. Askari F, Parizi MK, Jessri M, Rashidkhani B. Fruit and vegetable intake in relation to prostate cancer in Iranian men: a case-control study. Asian Pac J Cancer Prev 2014;15:5223-7.
16. Aune D, De Stefani E, Ronco A, Boffetta P, Deneo-Pellegrini H, Acosta G, Mendilaharsu M. Fruits, vegetables, and the risk of cancer: a multisite case-control study in Uruguay. Asian Pac J Cancer Prev 2009;10:419-28.
17. Subahir MN, Shah SA, Zainuddin ZM. Risk factors for prostate cancer in Universiti Kebangsaan Malaysia Medical Centre: a case-control study. Asian Pac J Cancer Prev 2009;10:1015-20.
18. Hurst R, Hooper L, Norat T, Lau R, Aune D, Greenwood DC, e Vieira R, Collings R, Harvey LJ, Sterne JA, Beynon R, Savović J, Fairweather-Tait SJ. Selenium and prostate cancer: systematic review and meta-analysis. Am J Clin Nutr 2012;96:111-22.
19. Etminan M, Takkouche B, Caamano-Isorna F. The role of tomato products and lycopene in the prevention of prostate cancer: a meta-analysis of observational studies. Cancer Epidemiol Biomarkers Prev 2004;13:340-5.
20. Wilson KM, Giovannucci EI, Mucci LA. Lifestyle and dietary factors in the prevention of lethal prostate cancer. Asian J Androl 2012;14:365-74.
21. Nguyen TT, Shaw PN, Parat MO, Hewavitharana AK. Anticancer activity of Carica papaya: a review. Mol Nutr Food Res 2013;57:153-64.
22. Rahmat A, Rosli R, Wan Nor I'zzah WMZ, Susi E, Huzaimah AS. Antiproliferative activity of pure lycopene compared to both extracted lycopene and juices from watermelon (Citrullus vulgaris) and papaya (Carica papaya) on human breast and liver cancer cell lines. J Med Sci 2002;2:55-8.
23. Nakamura Y, Yoshimoto M, Murata Y, Shimoishi Y, Asai Y, Park EY, Sato K, Nakamura Y. Papaya seed represents a rich source of biologically active isothiocyanate. J Agric Food Chem 2007;55:4407-13.
24. Udoh P, Essien I, Udoh F. Effects of Carica papaya (paw-paw) seeds extract on the morphology of pituitary-gonadal axis of male Wister rats. Phytother Res 2005;19:1065-8.
25. Panzarini E, Dwikat M, Mariano S, Vergallo C, Dini L. Administration dependent antioxidant effect of Carica papaya seeds water extract. Evid Based Complement Alternat Med 2014;2014:281508.
26. Li H, Parry JW. Phytochemical compositions, antioxidant properties, and colon cancer antiproliferation effects of Turkish and Oregon hazelnut. Food Nutr Sci 2011;2:1142-9.
27. Jiménez-Coello M, Guzman-Marín E, Ortega-Pacheco A, Perez-Gutiérrez S, Acosta-Viana KY. Assessment of the anti-protozoal activity of crude Carica papaya seed extract against Trypanosoma cruzi. Molecules 2013;18:12621-32.
28. He X, Ma Y, Yi G, Wu J, Zhou L, Guo H. Chemical composition and antifungal activity of Carica papaya Linn. seed essential oilagainst Candida spp. Lett Appl Microbiol 2017;64:350-4.
29. Lohiya NK, Manivannan B, Goyal S, Ansari AS. Sperm motility inhibitory effect of the benzene chromatographic fraction of the chloroform extract of the seeds of Carica papaya in langur monkey, Presbytis entellus entellus. Asian J Androl 2008;10:298-306.
30. Singh O, Ali M. Phytochemical and antifungal profiles of the seeds of carica papaya L. Indian J Pharm Sci 2011;73:447-51.
31. Pinto LA, Cordeiro KW, Carrasco V, Carollo CA, Cardoso CA, Argadoña EJ, Freitas Kde C. Antiulcerogenic activity of Carica papaya seed in rats. Naunyn Schmiedebergs Arch Pharmacol 2015;388:305-17.
32. García-Solís P, Yahia EM, Morales-Tlalpan V, Díaz-Muñoz M. Screening of antiproliferative effect of aqueous extracts of plant foods consumed in Mexico on the breast cancer cell line MCF-7. Int J Food Sci Nutr 2009;60 Suppl 6:32-46.
33. Morimoto C, Dang NH, DangYS. Cancer prevention and treating composition for preventing, ameliorating, or treating solid cancers, e.g. lung, or blood cancers, e.g. lymphoma, comprises components extracted from brewing papaya. Patent number WO2006004226-A1; EP1778262-A1; JP2008505887-W; US2008069907-A1 2008.
34. Otsuki N, Dang NH, Kumagai E, Kondo A, Iwata S, Morimoto C. Aqueous extract of Carica papaya leaves exhibits anti-tumor activity and immunomodulatory effects. J Ethnopharmacol 2010;127:760-7.
35. Pathak N, Khan S, Bhargava A, Raghuram GV, Jain D, Panwar H, Samarth RM, Jain SK, Maudar KK, Mishra DK, Mishra PK. Cancer chemopreventive effects of the flavonoid-rich fraction isolated from papaya seeds. Nutr Cancer 2014;66:857-71.
36. Jian L, Lee AH, Binns CW. Tea and lycopene protect against prostate cancer. Asia Pac J Clin Nutr 2007;16 Suppl 1:453-7.
37. Osiecki H. Cancer: a nutritional, biochemical approach. Eagle Farm, QLD: Bioconcepts Publishing; 2002.
38. Uche-Nwachi EO, Mitchell CV, McEwen C. Steroidogenic enzyme histochemistry in the testis of Sprague Dawley rats following theadministration the water extracts from Carica papaya seed. Afr J Tradit Complement Altern Med 2011;8:69-78.
39. Verma RJ, Nambiar D, Chinoy NJ. Toxicological effects of Carica papaya seed extract on spermatozoa of mice. J Appl Toxicol 2006;26:533-5.
40. Oderinde O, Noronha C, Oremosu A, Kusemiju T, Okanlawon OA. Abortifacient properties of aqueous extract of Carica papaya (Linn) seeds on female Sprague-Dawley rats. Niger Postgrad Med J 2002;9:95-8.
41. Yanty NA, Marikkar JM, Nusantoro BP, Long K, Ghazali HM. Physico-chemical characteristics of papaya (Carica papaya L.) seed oil of the Hong Kong/Sekaki variety. J Oleo Sci 2014;63:885-92.
42. Xu BJ, Chang SK. A comparative study on phenolic profiles and antioxidant activities of legumes as affected by extraction solvents. J Food Sci 2007;72:S159-66.
43. Shi J, Nawaz H, Pohorly J, Mittal G, Kakuda Y, Jiang Y. Extraction of polyphenolics from plant material for functional foods-engineering and technology. Food Rev Int 2005;21:139-66.
44. Soto-Hernandez M, Palma-Tenango M, Garcia-Mateos MR (eds). Phenolic Compounds - Biological Activity. Croatia: InTech; 2017.
45. Naggayi M, Mukiibi N, Iliya E. The protective effects of aqueous extract of carica papaya seeds in paracetamol induced nephrotoxicity in male wistar rats. Afr Health Sci 2015;15:598-605.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Alotaibi KS, Li H, Rafi R, Siddiqui RA. Papaya black seeds have beneficial anticancer effects on PC-3 prostate cancer cells. J Cancer Metastasis Treat 2017;3:161-8. http://dx.doi.org/10.20517/2394-4722.2017.33
AMA Style
Alotaibi KS, Li H, Rafi R, Siddiqui RA. Papaya black seeds have beneficial anticancer effects on PC-3 prostate cancer cells. Journal of Cancer Metastasis and Treatment. 2017; 3: 161-8. http://dx.doi.org/10.20517/2394-4722.2017.33
Chicago/Turabian Style
Alotaibi, Khalid S., Haiwen Li, Reza Rafi, Rafat A. Siddiqui. 2017. "Papaya black seeds have beneficial anticancer effects on PC-3 prostate cancer cells" Journal of Cancer Metastasis and Treatment. 3: 161-8. http://dx.doi.org/10.20517/2394-4722.2017.33
ACS Style
Alotaibi, KS.; Li H.; Rafi R.; Siddiqui RA. Papaya black seeds have beneficial anticancer effects on PC-3 prostate cancer cells. J. Cancer. Metastasis. Treat. 2017, 3, 161-8. http://dx.doi.org/10.20517/2394-4722.2017.33
About This Article
Copyright
Data & Comments
Data
 Cite This Article 13 clicks
Cite This Article 13 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.