Impact of previous anti-angiogenesis treatment in nivolumab-treated advanced non-small cell lung cancer
Abstract
Aim: To investigate how previous systemic therapy such as anti-angiogenesis can influence cancer immunotherapy for non-small cell lung cancer (NSCLC).
Methods: A total of 134 patients with advanced NSCLC who were treated with nivolumab were retrospectively reviewed. Correlation between status of prior anti-angiogenesis treatment and clinical characteristics were determined. Impact of prior anti-angiogenesis on therapeutic outcome of nivolumab was investigated for tumor efficacy such as progression-free survival (PFS).
Results: Sixteen patients were treated with at least one anti-angiogenesis agent prior to nivolumab. The prior use of anti-angiogenesis agent was associated with stage IV disease, non-squamous histology, and two or more lines of systemic therapy. Median PFS was significantly shorter in the prior anti-angiogenesis group than in no prior anti-angiogenesis group (8.3 vs. 11.3 weeks, log-rank P = 0.006). Multivariate analyses demonstrated that only prior anti-angiogenesis status was associated with worse PFS. There is also a slight trend for worse disease control rate (P = 0.101, Fisher’s exact test) and overall survival (P = 0.200, log-rank) in prior anti-angiogenesis group.
Conclusion: This retrospective study suggests that prior anti-angiogenesis treatment negatively impacts the therapeutic outcome of immunotherapy in advanced NSCLC.
Keywords
Introduction
Systemic treatment for advanced cancer had been primarily cytotoxic chemotherapy until modern systemic modalities were recently developed. Now we know that targeted therapies for selected advanced cancer such as oncogene-driven malignancy provide better outcomes than traditional chemotherapy. For instance, small molecule kinase inhibitors are available for advanced non-small cell lung cancer (NSCLC) with a somatic mutation in the catalytic domain of epidermal growth factor receptor gene (EGFR) or gene rearrangement in anaplastic leukemic kinase gene (ALK)[1-4]. More recently, inhibitors for immune checkpoints that negatively regulate anti-cancer immunity have become clinically available with improved survival outcome for the treatment of advanced NSCLC, head/neck, melanoma, bladder, and renal cell carcinomas[5-11].
The mortality rate for lung cancer, however, has not changed dramatically over the last several decades[12]. Although recently developed cancer immunotherapy, such as anti-PD-1 therapy, has made a significant impact on daily practice for advanced NSCLC, most patients who are treated with such agents still succumb to the disease within five years[13]. Continued efforts to enhance activity of cancer immunotherapy are required to further improve outcome.
Recently researchers have been conducting clinical trials to determine if the combination of immunotherapy and other treatments may have additive clinical activity in this disease. Anti-angiogenesis agents such as bevacizumab have been developed and achieved regulatory approval for several cancer types[14,15]. These agents are also being investigated in various diseases in combination with immunotherapy[16]. Rationale for the combination is that suppression of neoangiogenesis, remodeling on distorted microvasculature, and resultant improved tumor perfusion are expected to enhance anti-cancer immunity[16]. Because bevacizumab has a relatively long half-life (approximately 20 days) and lasting biological effect[15], previous anti-angiogenesis treatment might positively influence the efficacy of anti-cancer immunotherapy. Several studies have indicated that withdrawal of anti-angiogenesis agents results in an increase in tumor aggressiveness due to rebound angiogenesis in the tumor microenvironment[17,18]. We therefore conducted a retrospective study to determine if prior use of anti-angiogenesis therapy can impact progression-free survival in advanced NSCLC patients who were treated with anti-PD-1 therapy.
Methods
Patient selection
A total of 801 advanced and metastatic NSCLC patients were registered at University of Kansas Cancer Center between January 2015 and June 2016. Review of their medical records identified 141 patients who were treated with at least one dose of the anti-PD-1/PD-L1 inhibitors at University of Kansas Cancer Center. A majority (n = 133) of patients were treated with nivolumab alone, whereas others were treated with nivolumab and atezolizumab (n = 1), atezolizumab alone (n = 1), pembrolizumabalone (n = 4), or other investigational agentalone (n = 2). All of these agents were intravenously given every two weeks (nivolumab) or every three weeks (atezolizumab and pembrolizumab) according to standard dosing schedules.
Because most patients were treated with nivolumab (n = 134), we decided to focus on patients who received it for recurrent or metastatic disease. They were grouped based on presence or absence of previous anti-angiogenesis treatment which included bevacizumab and ramcirumab. None of the patients received other anti-angiogenesis agents prior to nivolumab. Information about clinical demographics was collected as well. The two groups (prior anti-angiogenesis vs. no prior anti-angiogenesis) were compared for the differences in clinical demographics and outcome. Tumor response was determined according to Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 criteria, and disease control rate (DCR) was defined as the sum of complete response, partial response, and stable disease rates. Due to retrospective analysis, repeat imaging to confirm response was not always performed. Progression-free survival (PFS) was determined by duration from the start of nivolumab to disease progression or death of any cause. Definition of disease progression for the purpose of determining PFS was based on RECIST 1.1 criteria and/or on the clinical grounds (i.e., clinical progression without formal radiologic assessment if patients were unable to perform re-staging).
Statistical analysis
The Kaplan-Meier curves were applied and the differences were assessed using the log-rank test. Univariate and multivariable Cox proportional hazard models were used in order to assess the effects of variable(s) on PFS of the patients. Association between anti-angiogenesis treatment and other clinical features were carried out using Chi-squared or Fisher’s exact test. JMP software version 14 (SAS Institute, Cary, NC, USA) was used to perform statistical analyses. For all statistical tests, significance was considered to be achieved when two-sided P value was less than 0.05. This study was reviewed and approved by University of Kansas Medical Center Institutional Review Board.
Results
Patient characteristics according to previous anti-angiogenesis treatments are shown in Table 1. Of the 134 patients who received nivolumab, the individual dose was 3 mg/kg or 240 mg flat for 30 and 104 patients, respectively. Sixteen patients received at least one dose of anti-angiogenesis agents prior to nivolumab. They were previously treated with bevacizumab alone (n = 11), ramucirumab alone (n = 4), or both (n = 1). The number of doses for anti-angiogenesis agents ranged between one and 13 with a median of six. In seven of those, no other systemic therapy was given between anti-angiogenesis regimen and nivolumab. Of the 134 patients, seven patients completed PD-L1 immunohistochemistry with tumor material. Only two were tested positive (≥ 1%).
Patient characteristics and correlation with previous anti-angiogenesis treatment (n = 134)
| Characteristics | Anti-angiogenesis n (%) | Total | P value | |
|---|---|---|---|---|
| Yes | No | |||
| Total | 16 (100) | 118 (100) | 134 | |
| Age | 0.173 | |||
| < 70 | 10 (63) | 92 (78) | 102 (76) | |
| ≥ 70 | 6 (37) | 26 (22) | 32 (24) | |
| Stage at diagnosis | 0.001 | |||
| III | 0 (0) | 45 (38) | 45 (34) | |
| IV | 16 (100) | 73 (62) | 89 (66) | |
| Histology | 0.002 | |||
| Nonsquamous | 15 (94) | 63 (53) | 78 (58) | |
| Squamous | 1 (6) | 55 (47) | 56 (42) | |
| Sex | 0.427 | |||
| Male | 11 (69) | 67 (57) | 78 (58) | |
| Female | 5 (31) | 51 (43) | 56 (42) | |
| ECOG Performance Status | 0.360 | |||
| 0-1 | 14 (88) | 103 (77) | ||
| 2+ | 2 (12) | 31 (23) | ||
| EGFR status | 1.000 | |||
| Positive | 0 (0) | 5 (4) | ||
| Negative/unknown | 16 (100) | 129 (94) | ||
| No. of nivolumab doses | ||||
| Range (median) | 1-35 (4.5) | 1-59 (5) | 0.208* | |
| Dose of nivolumab | 0.523 | |||
| 240 mg flat | 2 (12) | 28 (24) | 30 (22) | |
| 3 mg/kg | 14 (88) | 90 (76) | 104 (78) | |
| Reason for discontinuation | 0.408** | |||
| PD/Death | 15 (94) | 74 (63) | 89 (66) | |
| AE | 0 (0) | 8 (7) | 104 (78) | |
| Lost follow-up | 0 (0) | 5 (4) | 89 (66) | |
| Ongoing | 1 (7) | 30 (25) | 31 (23) | |
| Others | 0 (0) | 1 (1) | 1 (1) | |
| No. of systemic chemotherapy lines | < 0.0001 | |||
| 1 | 6 (38) | 103 (87) | 109 (81) | |
| 2+ | 10 (62) | 15 (13) | 25 (19) | |
As of June 10, 2017, a total of 31 patients are still being treated with nivolumab; one in the prior anti-angiogenesis and 30 in the no prior anti-angiogenesis group. Because of the inherent limitation of retrospective review, many patients were lost to follow-up after progression on nivolumab. Only six patients in the no prior anti-angiogenesis group received an anti-angiogenesis agent after progression on nivolumab, whereas none did in prior anti-angiogenesis group.
Patients in the prior anti-angiogenesis group had significantly higher likelihood of having stage IV disease, non-squamous histology, and two or more lines of systemic therapy prior to nivolumab as compared to the no anti-angiogenesis group. The difference in histology is expected because current regulatory approval for bevacizumab, which is used in most patients in this group, is indicated for only non-squamous NSCLC. There was no pseudoprogression in either group.
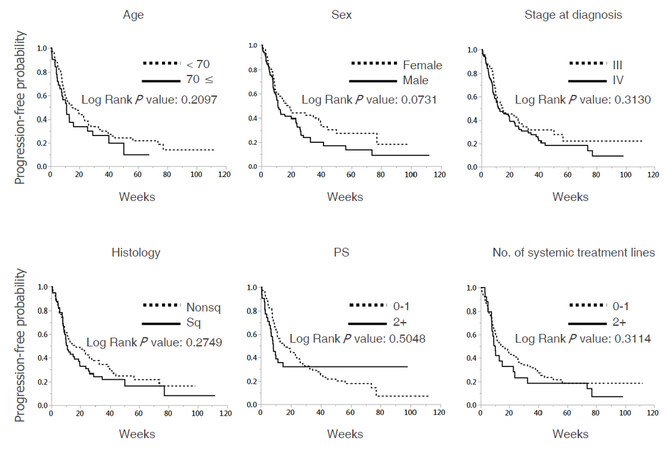
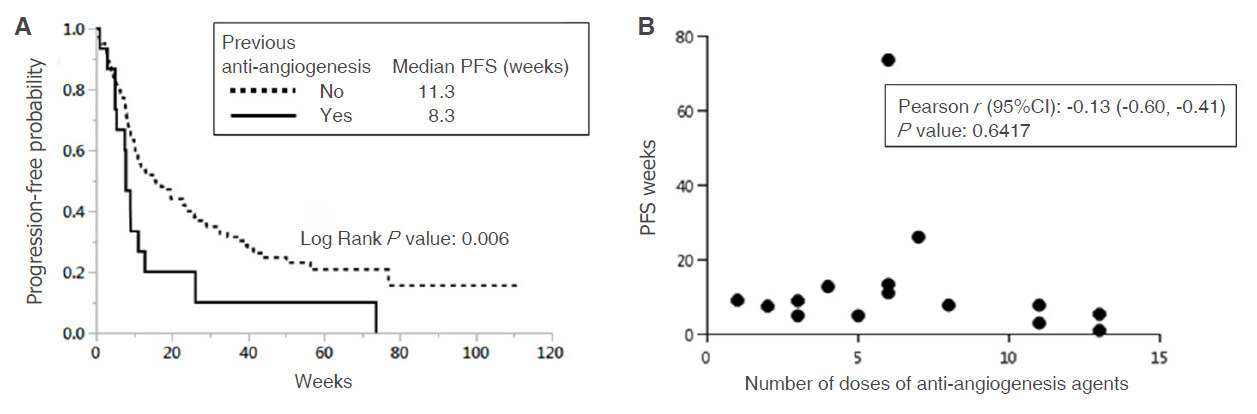
PFS and overall survival (OS) were investigated according to known prognostic factors as well as prior anti-angiogenesis status. Kaplan-Meier analyses demonstrated that the prior anti-angiogenesis group had a statistically shorter PFS as compared to the no prior anti-angiogenesis group, whereas no other factors demonstrated statistical difference (log rank P = 0.006, Figures 1 and 2A). Multivariate analysis for PFS showed that previous anti-angiogenesis remained statistically significant when other factors are being considered [Table 2]. There is no dose-response relationship between the number of doses of anti-angiogenesis agent and PFS [Figure 2B]. There is a trend in favor of the no anti-angiogenesis group in OS [Table 2] and DCR [Table 3], although the difference was not significant.
Figure 1. Progression-free survival according to clinical characteristics. Progression-free survival curves were plotted according to six clinical characteristics. log-rank tests were used for statistical analysis
Figure 2. Impact of previous anti-angiogenesis treatment on progression-free survival. A: Progression-free survival curves were plotted according to prior anti-angiogenesis treatment; B: Relationship between number of prior anti-angiogenesis doses and progression-free survival on nivolumab
Univariate and Multivariate analyses for prognostic factors on PFS and OS in NSCLC patients treated with nivolumab
| Factor | PFS | OS | ||
|---|---|---|---|---|
| Univariate analysis HR (95%CI) P value | Multivariate analysis HR (95%CI) P value | Univariate analysis HR (95%CI) P value | Multivariate analysis HR (95%CI) P value | |
| Age ( < 70 vs. ≥ 70) | 0.882 (0.698-1.136) | 0.938 (0.729-1.228) | 0.858 (0.659-1.146) | 0.952 (0.710-1.277) |
| 0.31909 | 0.633 | 0.288 | 0.744 | |
| Stage at diagnosis (III vs. IV) | 0.829 (0.652-1.038) | 0.849 (0.651-1.099) | 0.988 (0.766-1.258) | 0.959 (0.724-1.271) |
| 0.104 | 0.216 | 0.923 | 0.772 | |
| Histology (nonsquamous vs. squamous) | 0.939 (0.762-1.163) | 0.841 (0.659-1.077) | 0.788 (0.621-0.100) | 0.727 (0.549-0.964) |
| 0.561 | 0.168 | 0.050 | 0.027 | |
| Sex (female vs. male) | 0.810 (0.650-1.002) | 0.826 (0.660-1.026) | 0.829 (0.645-1.054) | 0.817 (0.636-1.048) |
| 0.052 | 0.084 | 0.127 | 0.107 | |
| Performance Status (0-1 vs. 2+) | 0.902 (0.710-1.172) | 0.877 (0.680-1.155) | 0.767 (0.591-1.019) | 0.716 (0.538-0.952) |
| 0.425 | 0.340 | 0.066 | 0.028 | |
| No. of systemic treatment (0-1 vs. 2+) | 0.848 (0.668-1.100) | 0.967 (0.733-1.303) | 0.893 (0.679-1.212) | 0.983 (0.705-1.371) |
| 0.205 | 0.820 | 0.451 | 0.919 | |
| Previous anti-angiogenesis (yes vs. no) | 1.466 (1.087-1.913) | 1.444 (1.009-2.029) | 1.277 (0.867-1.771) | 1.506 (0.979-2.316) |
| 0.014 | 0.044 | 0.200 | 0.074 | |
Best objective response according to prior anti-angiogenesis treatment
| Total n (%) | Prior anti-angiogenesis agent | P value | ||
|---|---|---|---|---|
| Yes | No | |||
| 134 (100) | 16 (100) | 118 (100) | ||
| ORR (CR + PR) | 11 (8) | 1 (6) | 10 (8) | 1.00 |
| Non-ORR | 123 (92) | 15 (94) | 108 (92) | |
| SD | 43 (32) | 2 (13) | 41 (35) | |
| PD | 40 (30) | 7 (44) | 33 (28) | |
| NE | 40 (30) | 6 (38) | 34 (29) | |
| DCR (CR + PR + SD) | 54 (40) | 3 (19) | 51 (43) | 0.101 |
| Others (PD + NE) | 80 (60) | 13 (81) | 67 (57) | |
Discussion
Discovery of immune checkpoints and development of agents to enhance T cell function has led to a drastic change in the management of advanced cancer, resulting regulatory approvals for several immunotherapy agents. Researchers are looking to potentiate T cell-mediated anti-tumor activity by adding agents with different mechanisms of action. For instance, cytotoxic chemotherapy, targeted agents, and anti-angiogenesis agents are being combined with anti-PD-1/PD-L1 inhibitors in ongoing clinical trials[16]. Except for one regimen which was recently approved via the accelerated approval process and still needing larger confirmatory studies[19], no combination regimen including immunotherapy is indicated for any human cancer. Patients with advanced cancer definitely require further development in systemic treatment which exceeds the current efficacy of single agent immunotherapy.
Targeting tumor neoangionegesis has been extensively investigated over the last few decades. Several agents have achieved regulatory approval in the treatment of advanced cancer[15,20-25]. In contrast to vascular endothelial growth factor receptor (VEGFR) kinase inhibitors for renal cell and hepatocellular carcinomas[20-23], monoclonal antibodies directed against VEGF/VEGFR are indicated for several cancer types only in combination with systemic chemotherapy[15,24]. For the treatment of advanced NSCLC, bevacizumab and ramucirumab are approved when combined with carboplatin-based regimens or docetaxel, respectively[15,24]. No anti-angiogenesis agent as monotherapy is indicated for NSCLC. Several studies with anti-angiogenesis agents have resulted in unexpected severe toxicity and a detrimental outcome for squamous NSCLC patients[26,27]. These findings indicate that anti-angiogenesis needs to be not only given in selected populations (i.e., non-squamous) but combined with agents with other mechanisms of action, because anti-angiogenesis by itself has only modest activity.
In addition to their modest clinical activity, the use of anti-angiogenesis agents in advanced cancer raised other concerns for researchers. Preclinical studies demonstrated that use and subsequent withdrawal of anti-VEGF agents could develop rebound tumor vascularization[17]. Others also reported induction of angiogenesis-related cytokines and epithelial-mesenchymal transition which enhance cancer invasiveness and eventual metastasis[28-31]. Clinical studies in patients with colorectal cancer also showed that continuation of bevacizumab beyond first progression was associated with prolonged overall survival, suggesting a detrimental withdrawal effect of anti-angiogenesis in humans as well[32].
Despite the abovementioned negative aspects for anti-angiogenesis agents, preclinical studies demonstrated therapeutic synergism between anti-angiogenesis and immunotherapy[33,34]. Targeting VEGF enhanced IFNγ-mediated upregulation of PD-L1 which in turn led to disease relapse in glioblastoma models. This negative effect of anti-angiogenesis treatment was nullified by dual blockade of the VEGF and PD-1/PDL1 signaling[34]. Supported by these preclinical observations, combination strategy using anti-angiogenesis agents and immune checkpoint inhibitors are actively tested in a number of clinical trials[35].
In this retrospective study, 16 (11.9%) of 134 patients who were treated nivolumab received anti-angiogenesis agents previously. This infrequent use of anti-angiogenesis agents in the first-line systemic therapy seems consistent with the study reported by Zhu et al.[36], where only 21.2% of stage IV NSCLC patients in their large SEER-Medicare analysis received bevacizumab in the first-line systemic therapy.
This study also showed that previous use of anti-angiogenesis agents was associated with significantly worse PFS. Overall response rate (ORR) and OS in the prior anti-angiogenesis group were also inferior to those in the no prior anti-angiogenesis group, although the differences were not statistically significant. Despite a relatively small number of patients in the prior anti-angiogenesis group, univariate and multivariate analyses demonstrated that prior anti-angiogenesis status is a poor prognostic factor independently for PFS. This detrimental effect of prior anti-angiogenesis on nivolumab treatment might be explained by withdrawal effect of anti-angiogenesis as discussed above. Consistent with this study, there are other similar clinical observations reported in the literature. A small retrospective study of 16 patients with glioblastoma reported a disappointing clinical effect when nivolumab was given after progression on bevacizumab[37]. A recent case series revealed that three patients with renal cell carcinoma with two or more lines of systemic anti-angiogenesis treatment developed rapid disease progression while on nivolumab treatment[38]. These patients received prior VEGFR TKIs prior to initiation of nivolumab. Moreover, in the pivotal phase III trial which led to Food and Drug Administration (FDA) approval of nivolumab for renal cell carcinoma, the difference in OS between the nivolumab and the control arms was not statistically significant when patients with two or more previous anti-angiogenesis agents were selected for subset analysis[11]. Although these observations, including this study, are still hypothesis-generating, the potential negative effect of prior anti-angiogenesis treatment warrants further investigation.
Retrospective observational studies such as this always have limitations. Various unappreciated biases exist in all retrospective studies. For instance, several patients in each group have never undergone formal re-staging but were considered as clinical progression which determined PFS. This single institution retrospective study needs to be validated by larger prospective and/or retrospective studies. Subset analyses within prior anti-angiogenesis group showed that there was no correlation between PFS and number of doses of anti-angiogenesis agents [Figure 2B] or interval from the last administration of anti-angiogenesis agent to first dose of nivolumab (data not shown). We acknowledge that these subset analyses require a larger sample size in order to establish clinical significance.
This retrospective study suggests that preceding anti-angiogenesis treatment has detrimental effect on subsequent treatment outcome of immunotherapy in NSCLC. This phenomenon might be associated with rebound tumor angiogenesis due to withdrawal of anti-angiogenesis treatment. This hypothesis needs to be confirmed by studies with a larger patient sample.
Declarations
AcknowledgmentsWe thank Tim Metcalf for providing administrative support at University of Kansas Cancer Center.
Authors’ contributionsConcept, design, clinical studies, data acquisition, data analysis: Komiya T
Literature search: Komiya T, Huang CH, Neupane P, Williamson SK
Statistical analysis: Komiya T, Chalise P
Manuscript preparation, manuscript editing, and manuscript review: Komiya T, Huang CH, Neupane P, Williamson SK, Chalise P
Data source and availabilityData and survey materials are available upon request from the corresponding author.
Financial support and sponsorshipNone.
Conflicts of interestThe authors have no conflict of interest.
Patient consentInformed consent was exempted by institutional IRB.
Ethics approvalAll procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Approval from institutional IRB was obtained.
Copyright© The Author(s) 2018.
REFERENCES
1. Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, Nishiwaki Y, Ohe Y, Yang JJ, Chewaskulyong B, Jiang H, Duffield EL, Watkins CL, Armour AA, Fukuoka M. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med 2009;361:947-57.
2. Zhou C, Wu YL, Chen G, Feng J, Liu XQ, Wang C, Zhang S, Wang J, Zhou S, Ren S, Lu S, Zhang L, Hu C, Hu C, Luo Y, Chen L, Ye M, Huang J, Zhi X, Zhang Y, Xiu Q, Ma J, Zhang L, You C. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol 2011;12:735-42.
3. Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, Felip E, Cappuzzo F, Paolini J, Usari T, Iyer S, Reisman A, Wilner KD, Tursi J, Blackhall F. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med 2014;371:2167-77.
4. Shaw AT, Kim TM, Crinò L, Gridelli C, Kiura K, Liu G, Novello S, Bearz A, Gautschi O, Mok T, Nishio M, Scagliotti G, Spigel DR, Deudon S, Zheng C, Pantano S, Urban P, Massacesi C, Viraswami-Appanna K, Felip E. Ceritinib versus chemotherapy in patients with ALK-rearranged non-small-cell lung cancer previously given chemotherapy and crizotinib (ASCEND-5): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol 2017;18:874-86.
5. Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE, Holgado E, Waterhouse D, Ready N, Gainor J, ArénFrontera O, Havel L, Steins M, Garassino MC, Aerts JG, Domine M, Paz-Ares L, Reck M, Baudelet C, Harbison CT, Lestini B, Spigel DR. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med 2015;373:123-35.
6. Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, Barlesi F, Kohlhäufl M, Arrieta O, Burgio MA, Fayette J, Lena H, Poddubskaya E, Gerber DE, Gettinger SN, Rudin CM, Rizvi N, Crinò L, Blumenschein GR Jr, Antonia SJ, Dorange C, Harbison CT, Graf Finckenstein F, Brahmer JR. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 2015;373:1627-39.
7. Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe S, O'Brien M, Rao S, Hotta K, Leiby MA, Lubiniecki GM, Shentu Y, Rangwala R, Brahmer JR. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 2016;375:1823-33.
8. Carbone DP, Reck M, Paz-Ares L, Creelan B, Horn L, Steins M, Felip E, van den Heuvel MM, Ciuleanu TE, Badin F, Ready N, Hiltermann TJN, Nair S, Juergens R, Peters S, Minenza E, Wrangle JM, Rodriguez-Abreu D, Borghaei H, Blumenschein GR Jr, Villaruz LC, Havel L, Krejci J, Corral Jaime J, Chang H, Geese WJ, Bhagavatheeswaran P, Chen AC, Socinski MA; CheckMate 026 Investigators. First-line nivolumab in stage IV or recurrent non-small-cell lung cancer. N Engl J Med 2017;376:2415-26.
9. Robert C, Long GV, Brady B, Dutriaux C, Maio M, Mortier L, Hassel JC, Rutkowski P, McNeil C, Kalinka-Warzocha E, Savage KJ, Hernberg MM, Lebbé C, Charles J, Mihalcioiu C, Chiarion-Sileni V, Mauch C, Cognetti F, Arance A, Schmidt H, Schadendorf D, Gogas H, Lundgren-Eriksson L, Horak C, Sharkey B, Waxman IM, Atkinson V, Ascierto PA. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med 2015;372:320-30.
10. Ning YM, Suzman D, Maher VE, Zhang L, Tang S, Ricks T, Palmby T, Fu W, Liu Q, Goldberg KB, Kim G, Pazdur R. FDA approval summary: atezolizumab for the treatment of patients with progressive advanced urothelial carcinoma after platinum-containing chemotherapy. Oncologist 2017;22:743-9.
11. Motzer RJ, Escudier B, McDermott DF, George S, Hammers HJ, Srinivas S, Tykodi SS, Sosman JA, Procopio G, Plimack ER, Castellano D, Choueiri TK, Gurney H, Donskov F, Bono P, Wagstaff J, Gauler TC, Ueda T, Tomita Y, Schutz FA, Kollmannsberger C, Larkin J, Ravaud A, Simon JS, Xu LA, Waxman IM, Sharma P; CheckMate 025 Investigators. Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med 2015;373:1803-13.
12. Cancer stat facts: lung and bronchus cancer. Surveillance, epidemiology, and end results. National Cancer Institute. Available from: https://seer.cancer.gov/statfacts/html/lungb.html [Last accessed on 10 Jan 2018].
13. Five-year survival rate for nivolumab-treated advanced lung cancer patients much higher than historical rate. AACR Annual Meeting; 2017 April 1-5, Washington, USA. Available from: http://www.aacr.org/Newsroom/Pages/News-Release-Detail.aspx?ItemID=1031#.WVFpd-srLIU [Last accessed on 10 Jan 2018].
14. Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, Lilenbaum R, Johnson DH. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med 2006;355:2542-50.
15. Highlights of prescribing information. AVASTIN® (bevacizumab). Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/125085s225lbl.pdf [Last accessed on 10 Jan 2018].
16. Manegold C, Dingemans AC, Gray JE, Nakagawa K, Nicolson M, Peters S, Reck M, Wu YL, Brustugun OT, Crinò L, Felip E, Fennell D, Garrido P, Huber RM, Marabelle A, Moniuszko M, Mornex F, Novello S, Papotti M, Pérol M, Smit EF, Syrigos K, van Meerbeeck JP, van Zandwijk N, Chih-Hsin Yang J, Zhou C, Vokes E. The Potential of combined immunotherapy and antiangiogenesis for the synergistic treatment of advanced NSCLC. J Thorac Oncol 2017;12:194-207.
17. Mancuso MR, Davis R, Norberg SM, O'Brien S, Sennino B, Nakahara T, Yao VJ, Inai T, Brooks P, Freimark B, Shalinsky DR, Hu-Lowe DD, McDonald DM. Rapid vascular regrowth in tumors after reversal of VEGF inhibition. J Clin Invest 2006;116:2610-21.
19. Langer CJ, Gadgeel SM, Borghaei H, Papadimitrakopoulou VA, Patnaik A, Powell SF, Gentzler RD, Martins RG, Stevenson JP, Jalal SI, Panwalkar A, Yang JC, Gubens M, Sequist LV, Awad MM, Fiore J, Ge Y, Raftopoulos H, Gandhi L; KEYNOTE-021 investigators. Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol 2016;17:1497-508.
20. Highlights of prescribing information. NEXAVAR® (sorafenib). Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/021923s008s009lbl.pdf [Last accessed on 10 Jan 2018].
21. Highlights of prescribing information. SUTENT® (sunitinib malate). Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/021938s13s17s18lbl.pdf [Last accessed on 10 Jan 2018].
22. Highlights of prescribing information. VOTRIENT® (pazopanib). Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/022465s-010S-012lbl.pdf [Last accessed on 10 Jan 2018].
23. Highlights of prescribing information. INLYTA® (axitinib). Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202324lbl.pdf [Last accessed on 10 Jan 2018].
24. Highlights of prescribing information. CYRAMZA® (ramucirumab).Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/125477s007lbl.pdf [Last accessed on 10 Jan 2018].
25. Highlights of prescribing information. ZALTRAP® (ziv-aflibercept).Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/125418s000lbl.pdf [Last accessed on 10 Jan 2018].
26. Johnson DH, Fehrenbacher L, Novotny WF, Herbst RS, Nemunaitis JJ, Jablons DM, Langer CJ, DeVore RF 3rd, Gaudreault J, Damico LA, Holmgren E, Kabbinavar F. Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol 2004;22:2184-91.
27. Scagliotti G, Novello S, von Pawel J, Reck M, Pereira JR, Thomas M, Abrão Miziara JE, Balint B, De Marinis F, Keller A, Arén O, Csollak M, Albert I, Barrios CH, Grossi F, Krzakowski M, Cupit L, Cihon F, Dimatteo S, Hanna N. Phase III study of carboplatin and paclitaxel alone or with sorafenib in advanced non-small-cell lung cancer. J Clin Oncol 2010;28:1835-42.
28. Ebos JM, Lee CR, Cruz-Munoz W, Bjarnason GA, Christensen JG, Kerbel RS. Accelerated metastasis after short-term treatment with a potent inhibitor of tumor angiogenesis. Cancer Cell 2009;15:232-9.
29. Shojaei F, Simmons BH, Lee JH, Lappin PB, Christensen JG. HGF/c-Met pathway is one of the mediators of sunitinib-induced tumor cell type-dependent metastasis. Cancer Lett 2012;320:48-55.
30. Ebos JM, Lee CR, Christensen JG, Mutsaers AJ, Kerbel RS. Multiple circulating proangiogenic factors inducedby sunitinib malate are tumor-independent and correlate with antitumor efficacy. Proc Natl Acad Sci U S A 2007;104:17069-74.
31. Lu KV, Chang JP, Parachoniak CA, Pandika MM, Aghi MK, Meyronet D, Isachenko N, Fouse SD, Phillips JJ, Cheresh DA, Park M, Bergers G. VEGF inhibits tumor cell invasion and mesenchymal transition through a MET/VEGFR2 complex. Cancer Cell 2012;22:21-35.
32. Grothey A, Sugrue MM, Purdie DM, Dong W, Sargent D, Hedrick E, Kozloff M. Bevacizumab beyond first progression is associated with prolonged overall survival in metastatic colorectal cancer: results from a large observational cohort study (BRiTE). J Clin Oncol 2008;26:5326-34.
33. Schmittnaegel M, Rigamonti N, Kadioglu E, Cassará A, Wyser Rmili C, Kiialainen A, Kienast Y, Mueller HJ, Ooi CH, Laoui D, De Palma M. Dual angiopoietin-2 and VEGFA inhibition elicits antitumor immunity that is enhanced by PD-1 checkpoint blockade. Sci Transl Med 2017;9:eaak9670.
34. Allen E, Jabouille A, Rivera LB, Lodewijckx I, Missiaen R, Steri V, Feyen K, Tawney J, Hanahan D, Michael IP, Bergers G. Combined antiangiogenic and anti-PD-L1 therapy stimulates tumor immunity through HEV formation. Sci Transl Med 2017;9:eaak9679.
35. Kanda S, Goto K, Shiraishi H, Kubo E, Tanaka A, Utsumi H, Sunami K, Kitazono S, Mizugaki H, Horinouchi H, Fujiwara Y, Nokihara H, Yamamoto N, Hozumi H, Tamura T. Safety and efficacy of nivolumab and standard chemotherapy drug combination in patients with advanced non-small-cell lung cancer: a four arms phase Ib study. Ann Oncol 2016;27:2242-50.
36. Zhu J, Sharma DB, Gray SW, Chen AB, Weeks JC, Schrag D. Carboplatin and paclitaxel with vs without bevacizumab in older patients with advanced non-small cell lung cancer. JAMA 2012;307:1593-601.
37. Chamberlain MC, Kim BT. Nivolumab for patients with recurrent glioblastoma progressing on bevacizumab: a retrospective case series. J Neurooncol 2017;133:561-9.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Komiya T, Huang CH, Neupane P, Williamson SK, Chalise P. Impact of previous anti-angiogenesis treatment in nivolumab-treated advanced non-small cell lung cancer. J Cancer Metastasis Treat 2018;4:1. http://dx.doi.org/10.20517/2394-4722.2017.65
AMA Style
Komiya T, Huang CH, Neupane P, Williamson SK, Chalise P. Impact of previous anti-angiogenesis treatment in nivolumab-treated advanced non-small cell lung cancer. Journal of Cancer Metastasis and Treatment. 2018; 4: 1. http://dx.doi.org/10.20517/2394-4722.2017.65
Chicago/Turabian Style
Komiya, Takefumi, Chao H. Huang, Prakash Neupane, Stephen K. Williamson, Prabhakar Chalise. 2018. "Impact of previous anti-angiogenesis treatment in nivolumab-treated advanced non-small cell lung cancer" Journal of Cancer Metastasis and Treatment. 4: 1. http://dx.doi.org/10.20517/2394-4722.2017.65
ACS Style
Komiya, T.; Huang CH.; Neupane P.; Williamson SK.; Chalise P. Impact of previous anti-angiogenesis treatment in nivolumab-treated advanced non-small cell lung cancer. J. Cancer. Metastasis. Treat. 2018, 4, 1. http://dx.doi.org/10.20517/2394-4722.2017.65
About This Article
Copyright
Data & Comments
Data
 Cite This Article 9 clicks
Cite This Article 9 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.