Overview of genetic signaling pathway interactions within cutaneous malignancies
Abstract
Melanoma and non-melanoma cutaneous malignancies are some of the leading causes of cancer-related death in the United States. Though melanoma is more known to have a high mortality rate, the total mortality per year is nearly equal for between melanoma and non-melanoma skin cancer. Moreover, the non-melanoma types of cutaneous malignancies have potential to become locally invasive and even metastasize with very little to no treatment options when advanced. The development of these malignancies involves various genetic pathways through the four hallmarks of cancer development: malignant cell growth, apoptosis evasion, the use of supporting stroma and vascularization, and modulating and promoting an inadequate immune response. The genetic signaling pathways of basal cell carcinoma, squamous cell carcinoma, verrucous carcinoma, basosquamous cell carcinoma, melanoma, and cutaneous T-cell lymphoma interact with each other through genetic predisposition as well as with environmental exposures. Furthermore, solar ultraviolet radiation and chronic inflammatory states are found to initiate the progression of many of these cutaneous malignancies. This paper includes validated models of genetic pathways, emerging pathways, and crosstalk between genetic pathways through the four hallmarks of cancer development. Moreover, unlike most reviews addressing oncogenetics of the well-recognized, as well as newly discovered, genetic pathway mutations, this review stresses that these pathways are not fixed but rather exist in dynamic, interrelated, interactive, complex, and adaptive flux states.
Keywords
Introduction
The molecular and genetic basis of the development of cutaneous malignancies involve multiple pathways which continue to evolve over time. Tumor initiation, promotion, and progression leading to the multiplication of abnormal cells are all effected by genetics, environmental factors, acute and chronic exposures, diet, trauma, and many other factors. Angiogenesis and metastasis are likely influenced by host dependent factors such as age and immunological status. Many skin malignancies environmental factors, such as UV radiation that causes photocarcinogenesis, are coupled with genetic and epigenetic alterations or extrinsic factors that contribute to local inflammation and dysregulation of normal pathways. Emerging evidence supports the role of chronic inflammation in skin carcinogenesis mediated by factors including nuclear factor-kappa B (NF-κB), signal transducer and activator of transcription 3 (STAT3), and hypoxia-inducible factor-1 alpha (HIF-1α). In this article, we review validated models including the hedgehog pathway for basal cell carcinomas, p53 (TP53) pathway for squamous cell carcinomas, and BRAF pathway for cutaneous melanomas, among others. Additionally, we address emerging pathways that have not been completely elucidated including those implicated in cutaneous T-cell lymphoma. We also review the presumptive biology of less common skin cancers including basosquamous cell carcinoma and verrucous carcinoma.
In this review we approach the role of genetic events in relationship to four interactive processes referred to as “cancer hallmarks”. These signals must interact for cells which have undergone a carcinogenic event to survive, proliferate, maintain a footing, and spread. The four hallmarks we review are: (1) malignant cell growth; (2) prevention of apoptosis; (3) promoting use of supporting stroma and vascularization; and (4) modulating and promoting an inadequate immune response. The genetic pathways of cutaneous malignancies will be grouped as related to basal cell carcinoma, squamous cell carcinoma, verrucous carcinoma, basosquamous cell carcinoma, melanoma, and cutaneous T-cell lymphoma. Nonetheless, this review differs from most discussions concerning oncogenetics because we stress that these well-recognized, as well as newly discovered, genetic pathway mutations are not fixed but rather exist in dynamic, interrelated, interactive, complex, and adaptive flux states.
Genetics of basal cell carcinoma
Nonmelanoma skin cancers (NMSC) include cancers affecting keratinocytes such as basal cell carcinoma (BCC) and squamous cell carcinoma (SCC). This term also includes Merkel cell carcinoma (MCC), a rare neuroendocrine tumor, and cutaneous T-cell lymphoma (CTCL). Four interactive processes referred to as the four hallmarks must interact for cells which have undergone a carcinogenic event to survive, proliferate, maintain a footing, and spread.
Malignant cell growth
Ultraviolet radiation
The most prevalent carcinogenic event promoting NMSC is ultraviolet radiation (UV) from sun exposure and/or from other UV sources such as tanning beds[1]. Skin damage from sun exposure modulates tumorigenesis by damaging DNA, creating an inflammatory state, and activating oxidative stress responses, receptor tyrosine kinases (RTK), and pro-apoptotic pathways[2]. UV, a recognized carcinogen, penetrates through the skin and damages keratinocytes of the basal epidermal layer. UV is emitted from the sun as well as from some man-made light. UVA (315-400 nm) compared to UVB (280-315 nm) accounts for 95% of sunlight. UVA is the primary source of light in tanning beds and it is less mutagenic[3,4]. UVA penetrates deeper into the epithelium affecting dermal stroma, while UVB is absorbed into the stratum corneum layer[5]. Primarily, UV causes DNA damage by generating cyclobutene pyrimidine dimers and 6-4 photoproducts[6]. Under stress, Sestrin2, an antioxidant, is activated by tumor suppressor p53 and inhibits positive cell growth regulator mammalian target of rapamycin (mTOR)[7,8]. The activity and skin penetration of UV depends on its wavelength with UVA penetrating between 700 nm - 1 mm and UVB penetrating between 280-320 nm[9].
Damage to chromosome 9 hedgehog pathway
BCC is the most common NMSC and accounts for 80% of NMSC diagnoses[10]. BCC-specific risk factors include intermittent/recreational sun exposure, other sources of UV light, ionizing radiation, and skin phototype[11]. The most critical pathway in BCC tumorigenesis appears to be the Hedgehog [sonic hedgehog (SHH)] pathway composed of three genes: SHH, DHH, and IHH as well as two patched genes: PTCH1 and PTCH2. The SHH pathway is important for the patterning, growth, and development of vertebrates. The patched genes encode SHH pathway receptors responsible for suppressing Smoothened (SMO), a transmembrane protein/proto-oncogene capable of activating Glioma-associated oncogene (Gli) transcription factors[12]. In the SHH pathway, a SHH ligand binds to its receptor, PTCH (a transmembrane protein) to disinhibit SMO[13]. The activation of SMO, which occurs in the primary cilium, causes the accumulation of intracellular calcium ions resulting in a disruption in calcium homeostasis[14,15]. The transcription of Gli proteins has been shown to be sufficient to induce BCC development[16]. After the SHH pathway, TP53 gene point mutations are the second most common genetic mutation in BCCs[11,17]. Traditional advanced BCC therapy consists of surgical resection, but the advent of small-molecule inhibitors of the SHH pathway allowed for new therapeutic options for patients with locally advanced or metastatic BCC[13,18].
Activation of the SHH pathway at the level of PTCH involves SHH interaction with PTCH through two distinct interfaces, the interface between PTCH and the calcium and zinc binding surfaces of SHH and the interface between PTCH and the N-terminal palmitoyl and C-terminal cholesteroyl moiety of SHH. Mutations at these interfaces that increase SHH binding to PTCH may subsequently increase the signaling strength of the SHH pathway and may drive tumorigenesis in BCC. Activation of the SHH pathway at the level of SMO involves side-chain oxysterols (endogenous cholesterol metabolites) that induce SMO accumulation in primary cilia even in the absence of SHH ligands. Excess side-chain oxysterols may lead to overactivation of the SHH pathway while pharmaceutical and genetic approaches aimed at reducing cellular cholesterol levels have been shown to attenuate SHH signaling in target cells, highlighting a potential role of excess side-chain oxysterols in the pathogenesis of BCC[19][Figure 1].
DNA damage repair
Damaged keratinocytes must depend on DNA repair mechanisms such as ataxia-telangiectasia-mutated (ATM) and ataxia telangiectasia and Rad3-related (ATR) proteins, members of the PIKK family of proteins. After the cell experiences a UV-induced double-stranded DNA break, ATM undergoes autophosphorylation which empowers ATM to phosphorylate checkpoint kinase (CHK) 2[20,21]. CHK2 inhibits CD25 phosphatases which then prohibit the cell from undergoing mitosis[22]. ATM also can phosphorylate p53, arresting the cell in G1[23]. If the UV induces a single-stranded DNA break, ATR will be activated. ATR will autophosphorylate and then CHK1 which goes on to phosphorylate CDC25[24,25]. This, once again, prevents the cell from completing mitosis. In addition, ATR is involved in the p21 pathway which can modulate various cyclins/CDKs to inhibit the cell cycle[26]. ATR phosphorylates murine double minute 2 (MDM2) to then inactivate p53. The inactivation of p53 results in the uncontrolled proliferation of cells with damaged DNA. On the other hand, ATR and ATM can directly phosphorylate E3 ubiquitin ligase SIAH1 which activates p53. The activation of p53 sends cells that normally should be permitted to proliferate to go into apoptosis. Thus, the dysregulation of ATR and ATM results in unpredictable cell cycle activity, especially at the p53 checkpoint which is known as “the guardian of the genome”.
Apoptosis evasion
Role of UV
UV exposure activates stress responses in the epidermis such as reactive oxygen species production which damages lipids, proteins, and DNA while also invoking antioxidant defense by suppressing tumorigenesis and initiating apoptotic pathways[27]. The conflicting evidence forces the consideration that antioxidative therapies may be counterintuitive in the treatment of certain cancers. When observing the tumors of melanoma and non-melanoma patients, NMSC had lower levels of superoxide dismutase and catalase when compared to the melanoma samples. This observation suggests that NMSC are associated with weaker antioxidative defenses during tumorigenesis[28]. To examine the signaling involved in oxidative stress, we will explore the p38 mitogen-activated protein kinases (p38) and c-Jun N-terminal kinases (JNK) pathways, both of which have been shown to be involved in pro- and anti-apoptotic mechanisms[29].
P38 signaling
P38 is a Raf-mitogen activated protein kinase (MAPK) protein that can respond to oxidative stress by triggering apoptosis. When oxidative stress activates the p38 system in keratinocytes, apoptosis signal-regulating kinase 1 may become activated[30]. Oxidative stress also inhibits MAPK phosphatases resulting in increased p38 activation[31]. UV alone suffices in activating the p38 signaling pathway[30,32].
JNK signaling
JNK is another member of the MAPK family that can be activated in as little as 5 min post-UV exposure. JNK targets activator protein-1 (AP-1) which is an oncogenic transcription factor involved in cell cycle regulation[33]. Although associated with pro-apoptotic activity, AP-1 can promote survival through crosstalk with the NF-kB pathway. Pharmacological studies have shown that the inhibition of JNK in human keratinocytes in vitro results in greater UV-induced apoptosis[34]. The same effect was shown in vivo when inhibiting AP-1 in dominant negative c-jun hairless mice[35].
Stroma vascularization effects
RTK activation
RTKs are a group of receptors activated in response to UV exposure[36]. Some of the more well-known RTKs include the IGF1-R, EGFR, FGFR, and VEGFR[37]. RTK activation and RTK downstream pathways are targets for drug development[38]. In NMSC, cetuximab (an EGFR inhibitor) is available as a pharmaceutical therapy[39]. There are two main RTK activation pathways in NMSC, PI3K/mTOR signaling and RAF/MEK/ERK signaling[37][Figure 2].
PI3K/mTOR signaling
There are two forms of mTOR, mTORC1 and mTORC2, and both forms are involved in the development of NMSC secondary to sun exposure[40]. PI3K and tensin homolog (PTEN) mutations are common in NMSC[41,42]. To activate mTORC1, PI3K is recruited to the receptor which then leads to the phosphorylation of the p85 subunit. This allows PI3K to phosphorylate PIP2, converting it to PIP3. PIP3 then recruits phosphoinositide-dependent kinase 1 and AKT to the receptor to activate AKT. AKT then phosphorylates the MTORC1 negative regulator which leads to the activation of mTORC1, signaling for cell growth and proliferation[43,44][Figure 3].
Figure 3. mTORC1 and two signaling pathways. This figure describes the transcriptional mTORC1 and mTORC2 pathways
Activation of mTORC2 occurs once PI3K phosphorylates PIP2 to PIP3[45]. Part of the SIN1 protein is recruited to the cell membrane where it binds to PIP3, resulting in a conformational change in mTORC2 that reveals an active kinase site[24]. PIP3 then recruits AKT to mTORC2 which results in the phosphorylation of AKT at the active kinase site[46].
FOXO3 signaling
The FOX”O” family of proteins are transcription factors known to regulate tumor longevity and suppression[47,48]. FOXO3a transcriptionally targets apoptotic genes such as FasL, TNF-related apoptosis-inducing ligand gene (TRAIL), BIM, and PUMA, all of which are involved in UV-induced apoptosis[49-51]. AKT is a negative regulator of FOKO3a that acts by translocating into the cell nucleus with a chaperone protein, 14-3-3[52]. Together, AKT and its chaperone reduce FOXO3a activity, sensitizing keratinocytes to UVB-induced apoptosis[49-51,53].
Raf/MEK/ERK signaling
The Raf/MEK/ERK pathway can be activated secondary to UV exposure. Raf binds MEK1 and MEK2, phosphorylating them. MEK1 and MEK2 then phosphorylate ERK[54]. ERK1 is involved in the regulation of cell growth, malignant transformation, and drug resistance[55]. A study found that cells treated with SMO inhibitors (a hedgehog pathway BCC treatment) have a tendency to increase RAS/MEK/ERK signaling, leading to the development of SCC[56]. Thus, therapies should consider targeting the mTORC2/AKT and RAF/MEK/ERK pathways to simultaneously suppress BCC and SCC malignancies[57].
Isoelectric signaling
There appears to be a relationship between the isoelectric signaling and gene modulation. One study showed that IFN-γ can lead to the loss of EGF’s inhibition of basolateral K+ channel activity[58]. IFN-γ levels are often elevated during periods of inflammation which leads to the shift in signaling to favor the EGF-stimulated pathways (such as Raf/MEK/ERK and PI3K/mTOR described above)[59].
Modifying immune response
NF-κB pathway
Aside from the direct effects of sunlight, skin injury resulting in inflammatory response may also lead to tumorigenesis in NMSC. When the skin is damaged, such as in UV-induced sunburns, the microenvironment of the damaged area changes to allow for the extravasation of leukocytes to the tissue injury site. NFκB is an established pathway in the mediation of inflammation. NFκB is a heterodimer of p65 and p50 subunits who are both bound to IkB, an inhibitory protein. Toll-like receptors, tumor necrosis factor (TNF) receptors, and RTK can all cause NFκB activation by activating IkB via phosphorylation[60]. This process removes the inhibition of NFκB, allowing the protein to move into the nucleus and promote the transcription of pro-inflammatory genes such as TNF-α, IL-1 IL-6, IL-8, and various other cytokines and interferons[61]. Furthermore, p65-dependent NFκB signaling nurtures a pro-inflammatory environment inducing SCC tumor initiation and promotion[60,61].
STAT3 pathway
Pro-inflammatory growth factors and cytokines induce the expression of the STAT family of transcription factor. STAT3 enhances the transcription of factors related to inflammation, tumor promotion, cell survival, and metastasis[62]. In mice exposed to UVB, the overexpression of STAT3 results in accelerated skin tumorigenesis whereas the removal of STAT3 genes confers a resistance to skin tumor formation[63].
Once thought of as independent determinants of tumorigenesis, the four hallmarks of cancer form an interdependent network of signals that promote successful tumor growth. The study of skin cancer occurs under various conditions and laboratory settings. The high degree of variability in models, UV dosage, dimensions (2D vs. 3D), and methodologies creates potential confounds in the comparison of cancer signaling pathway.
Genetics of squamous cell carcinoma
Cutaneous squamous cell carcinoma (cSCC) is one of the most common forms of skin cancer worldwide. cSCCs most commonly present on the head and neck, particularly on the forehead, face, ears, and cheeks. Although melanoma is categorically perceived as a higher threat in society, more than 15,000 patients die from metastatic cSCC each year in the United States; this figure exceeds the total mortalities attributed annually to melanoma[64]. In addition, the mortality rate of metastatic cSCC is over 70%[65]. While multiple treatment modalities have been developed for advanced melanoma, advanced cSCC continues to have poor prognosis and limit options for treatment besides a newer immune checkpoint inhibitor, Cemplimab[66].
The factors evoking the carcinogenesis of cSCC are multiple and varied; the primary contributor is exposure to solar UV radiation. cSCC may also be initiated by industrial and inorganic exposures to tar, crude paraffin oil, fuel oil, creosote, lubricating oil, nitrosoureas, and arsenic in medications, foods, and drinking water[67]. Organ transplant recipients and other immunosuppressed patients have increased risk for cSCC, especially locally advanced cSCC (lacSCC) and metastatic cSCC. Additionally, cSCC can develop in the setting of chronic inflammation. For example, cSCCs may develop from chronic ulcers, psoriasis, cutaneous lupus erythematosus, radiation dermatitis, porokeratosis, and lichen sclerosus. Understanding the various genetic pathways in which cSCC emerges will provide insight into improved and individualized treatments of cSCCs. The underlying genetic processes involved in the initiation, promotion, maintenance, and establishment of aggressive growth patterns resemble the pathophysiology hallmarks common to all cancers: cell growth, prevention of apoptosis, development of supportive stroma and vascularization, and modulation of the immune response.
Thus, each of the carcinogenic pathways for cSCC will follow complex genetic crosstalk between pathways for each of these hallmark systems as well as between these components. These signaling pathways may be unique to a hallmark component of the genetic pathway or be present in multiple parts of the genetic pathway. These genetic pathways include primary, well recognized, genetic mutations of signaling pathways, initiation of alternative genetic pathways, or modulation to and between alternative genetic pathways. Each of these signaling pathways may vary in how it affects the overall status of cSCC through one or more hallmarks systems.
UV radiation
Cell growth
The EGFR pathway is important for keratinocyte proliferation, turnover, and wound repair. In normal cells, EGF, transforming growth factor-α (TGF-α), TGF-β, and platelet derived growth factor (PDGF) are four of many ligands that must bind to EGFR for its activation. After binding, the receptor undergoes dimerization and autophosphorylation, followed by the signaling proteins with the SH2 domain attaching to the Grb2 adapter protein and SOS complex[68]. This cascade of events continues with phosphorylation of the Ras/Raf/Map kinase pathway which then activates mTOR pathways[69]. This cascade of events is similar to that described in the BCC section. mTOR causes a cascade of signaling events that culminate in combinations of DNA replication, protein synthesis, and lipogenesis. Hyperproliferative lesions, including psoriatic plaques, seborrheic keratoses, and cutaneous malignancies, have been found to have mutations in EGFR[70]. Often this is due to the uncontrolled activation of the receptor without the necessary ligands present (i.e., EGF, TGF-α and β, PDGF). In addition, UV mutates the EGFR, promoting uncontrolled keratinocyte replication and eventually cSCC showing that these chronic inflammatory conditions also have malignant potential [Figure 2]. The similarities in pathway mutations between BCCs and cSCC demonstrate that BCCs and cSCCs have the potential to have modulating and communicating pathways. Rarely discussed, is this concept that the homeostatic flux of signals is imbalanced, allowing malignant keratinocytes to behave like benign keratinocytes through the option to differentiate into a BCC or cSCC.
Genetic crosstalk
In addition, crosstalk between these genetic events plays a marked role in cSCC. The EGF ligand promotes the activation of the EGFR pathways stimulating wound repair and eventually promotes hyperproliferative disease if uncontrolled. In addition, the ligands that activate EGFR can also activate the SHH pathway. It has been found that some BCCs have the potential of mutating to SCCs. The exact pathophysiology behind this event will be discussed in the basosquamous cell carcinoma section. Given that the mutation of the EGFR signaling pathway is known for producing SCCs, this pathway also has crosstalk with the SHH pathway, recognized as a culprit for the growth of BCCs. The pathway has been shown to increase intracellular calcium through activation of protein kinase C and MMPs, which incites further EGFR pathway activation[71][Figure 4]. However, the extracellular environment found within the mouse embryonic stem cells could be a hypercalcemic environment. A hypocalcemic environment may cause an imbalance within the SHH pathway leading to atypical signaling and activation of other modulating receptors[71][Figure 5]. Because the crosstalk between the EGFR pathway and SHH pathway occurs early in the activation of the SHH pathway, the blockade of SHH can lead to shunting of the SHH pathway to the EGFR pathway causing upregulation in the EGFR pathway and increased proliferation of keratinocytes. Eventually, these tumors have the potential to mutate to a different type of skin cancer [Figure 4][72].
Figure 4. Crosstalk with SHH pathway: this image describes the crosstalk between multiple pathways. SHH: sonic hedgehog; SMO: smoothened; TGF: transforming growth factor
Figure 5. SHH pathway increases intracellular calcium, which can also increase the activation of EGFR. SHH also activates MMPs, which will activate the EGFR and increase cellular growth[71]. SHH: sonic hedgehog; EGFR: epidermal growth factor receptor; MMP: metalloproteinases; NF-κB: nuclear factor-kappa B
Moreover, NOTCH gene mutation contributes to cSCC development through its role in chronic inflammation, trauma, or both. NOTCH genes are responsible for the production of NOTCH proteins involved in cell proliferation and regulation of apoptosis[73]. Cytochrome P450 family 1 subfamily B member 1 (CYP1B1), a heme-thiolate monooxygenase, is involved in estrogen metabolism and biosynthesis as well as a catalyst to hydroxylation of E2 to 4-hydroxyestradiol. Normal keratinocytes have been found to express this enzyme. In fact, recent studies have found that CYP1B1 antagonizes the signaling of NOTCH1, which, in turn, blocks keratinocyte proliferation and differentiation. Western blot and immunofluorescence display that there is increased involucrin, keratin 10, and ki67 (a proliferation marker) after downregulation and knock out of CYP1B1, demonstrating augmentation of keratinocyte proliferation and differentiation[74]. More research is being performed on the effect of CYP1B1 and NOTCH1 within SCC cells. This innovative research can lead to a new creation of a pharmacologic agent that can treat lacSCC in the future[74].
Other NOTCH pathway genes include the recombination signal binding protein for immunoglobulin J gene (RBPJ). This gene codes for RBP-Jκ also known as “CBF1, Suppressor of Hairless, Lag-1” (CSL) transcription factor within keratinocytes. Upper epithelial cells downregulate the production of CSL. However, pre-malignant and cSCCs demonstrate increased levels of CSL, leading to the excessive proliferation of normal epithelial cells underlying the development of actinic keratoses and cSCCs[75].
Role of microRNAs
MicroRNAs (miRNAs) play an important role in cell growth of cSCCs. However, the function of the many miRNAs varies. Within various signaling pathways, some miRNAs are upregulated, while others are downregulated, causing various responses. However, all these miRNAs increase cell growth of cSCC malignant lesions. For example, miR-21 works as an oncogene that targets transcription factor GRHL3, an important element in the PTEN pathway. When miR-21 is upregulated, the PTEN and PI3K/Akt/mTOR signaling pathways are overexpressed leading to increased cell growth[76]. This is one of the many miRNAs that influence cell growth of cSCCs. In fact, some of the upregulated miRNAs determine the aggressiveness of the cSCC lesion. Because miRNAs influence both the PTEN and mTOR pathways, this information always shows potential of crosstalk between the BCC and cSCC pathways.
Apoptosis evasion
An important cause of DNA instability is solar UV. Both UVA and UVB radiation induce the most common types of cSCC. It is well-established that UVB radiation causes direct keratinocyte DNA damage leading to cSCC, but exposure to UVA radiation has also been shown to foster tumorigenesis through DNA damage resulting from photosensitizers within the body causing indirect production of ROS. In natural sunlight, the ratio of UVA:UVB varies depending on the day, season, and latitude[77]. It is not well understood how exposure to variable ratios of UVA:UVB radiation affects progression through these oncogenic pathways. However, use of sunscreen that blocks UVB without blocking UVA, as well as chronic or acute UV exposure through window glass, can possibly increase the ratio. Foods containing furocoumarins (psoralens) also may increase the absorption of UVA and thus alter the UVA:UVB ratio[78]. Moreover, patients should be precautioned that substances applied to the skin such as retinoids, tanning oils and products with methyl and benzyl nicotinate may have the opposite effect of sunscreen and enhance the cutaneous absorption of UV radiation[79].
Solar UV radiation can induce DNA mutations within the cyclobutene pyrimidine dimers and 6-4 photoproducts[80]. These mutations can activate the ataxia telangiectasia and Rad3 (ATR) DNA repair system that, in turn, activate TP53, a tumor suppressor important for apoptosis. The BCC sections elaborate on the mechanisms of ATR. Mutation in either or both genes can lead to apoptotic escape and resultant overactive cell growth.
Mutation of cutaneous TP53 thus evokes an uncontrolled activation of the cell cycle allowing mutated cells to progress to the synthesis phase (S phase) of the cell cycle prematurely. Wildtype CDKN2A/p16 is responsible for generating cyclin dependent kinase (cdk) inhibitors INK4A and p14, both of which are TP53 degradation inhibitors. INK4A binds to cdk4 and cdk6, inhibiting cell cycle progression into the S phase. When the oncogene CDKN2A/p16 is mutated, INK4A is mutated, resulting in a lack of inhibition of cdk4 and cdk6 within the cell cycle. This mechanism resembles that of TP53 in that the cell cycle prematurely progresses into the S phase allowing unregulated DNA replication and advancement into the mitotic phase of the cell cycle. Recent studies have discovered with flow cytometry targeting ATR-activated cells can eradicate the number of cells with 6-4 photoproducts. Eliminating these cells can block solar UV radiation-induced damage from occurring and potentially cSCC tumorigenesis [Figure 6].
Figure 6. Cell cycle displaying TP53 inhibition of cell proliferation. G1 to S phase by CDKN2A blockade of cdk4 and cdk6[81]
Downregulation of certain miRNAs can affect apoptosis; specifically, the downregulation of miR-34a decreases the function of TP53 leading to avoidance of apoptosis. Select solar UV radiated keratinocytes appear to have the downregulation of miR-34a, leading to the proapoptotic effect of these tumor cells[76].
Keratoacanthomas (KA) are controversial lesions. These lesions are classified as cSCCs or lesions with cSCC potential that can spontaneously resolve. Nevertheless, taking the risk of waiting for a KA to spontaneously resolve can be detrimental, since not all lesions regress. Some KAs continue to grow rapidly; a subset may become aggressive and metastasize. There are many gene mutations that cause the transformation of signaling pathways that eventually lead to the appearance of these lesions. The most common type of genetic pathway that is mutated is the apoptotic pathway. The most noteworthy upregulated genes found through microarray analysis in KAs were MALAT-1, S100A8, and EHF. These genes modulate caspases, Bax, and Bcl-2, leading to avoidance of apoptosis[82]. Microarrays show thousands of 1449 genes that vary from those of cSCC, yet there are many similar gross and histological features between cSCC and KA. Regardless of this debate, KAs have a highly malignant potential similar to those of cSCC and should be treated as soon as they are discovered.
Immune response evasion
UVB radiation (and to a lesser extent, UVA radiation) modulates the immune response over time. Actinic keratosis (AK) is a pre-cancerous lesion that may evolve into cSCC. Whether AK is to be viewed as a dysplastic, precancerous lesion of keratinocytes or if they should rather be understood to be highly scattered, disorganized cSCC in situ has been contested in the realms of dermatopathology and histopathology[83]. Further research will be required to elucidate the proper classification of AKs. For this discussion, AKs will be considered precancerous lesions. The progression from AK to cSCC is associated with UVA and UVB modulation of immune signaling pathways. Chronic UV radiation causes an increase in p53 associated inflammation affecting apoptosis of keratinocytes; however, only roughly 10% of AKs progress to cSCC. In one school of thought, the fate of AK is dependent a stepwise progression through several stages, driven by complex immunologic mechanisms. In many cases, AKs may present asymptomatically with little or no inflammation. These lesions may be subclassified as asymptomatic actinic keratoses (AAK). Although initially asymptomatic, some AKs become inflamed and present symptomatically and thus may be classified as inflamed actinic keratoses (IAKs).
It is thought that some AAKs progress to IAKs as cells undergo molecular and immunologic changes that promote increased growth and inflammation. For example, Fas ligand (FasL) site desensitization may lead to the inhibition of CD8+ T lymphocytes resulting in impaired CD8+-mediated apoptosis and thus promote lesion progression[76]. Other AAKs may not undergo the changes necessary for progression and remain asymptomatic. It has also been shown that IAKs recruit immune cells, including CD3+, CD4+, CD8+ T lymphocytes, and Langerhans cells, producing further inflammation[84]. Should an IAK experience sufficient growth and keratinocyte damage, it may progress to cSCC in situ or Bowen’s Disease. Bowen’s Disease is slow-growing and shows displays only partial-thickness atypia. With further inflammation, damage, and mutations of keratinocytes, Bowen’s disease can evolve into cSCC with full thickness atypia and potential to invade and metastasize. The role of inflammation in the progression from AAK to IAK to cSCC is also supported by a stepwise increase in the expression of Bcl-2. Overall, the UV damage of keratinocyte DNA leads to the progression of a pre-cancerous asymptomatic lesion into a malignancy.
Secondary to UV induced cSCC are those induced through keratinocyte signaling mutation induced by chronic inflammation. Injury to the skin causes a wound healing cascade to occur, activating keratinocyte proliferation and many of the cell signaling pathways that are involved in tumorigenesis. The EGFR pathway, the intrinsic pathway, and the extrinsic inflammatory pathways are all included in the cell signaling pathways causing tumorigenesis. Chronic inflammatory conditions, including lichen sclerosus, may activate the intrinsic pathways causing chronic inflammation, increased cell turn over, and possible tumorigenesis by activating wound healing and repair simultaneously[85]. The extrinsic pathway includes the influence of environmental factors and viral causes of inflammation, including human papillomavirus (HPV)[85][Figure 7]. The viral mechanisms of HPV will be discussed later in the verrucous carcinoma section. This pathway involves the release of a cascade of inflammatory cytokines and chemokines which promote the migration of lymphocytes to the injury site and subsequent repair. Chemokines can enhance cell turnover causing increased DNA replication, predisposing cells to genetic mutations that foster the formation of tumors such as cSCC. In addition, cutaneous unilateral linear porokeratosis, another chronic inflammatory disease, has high potential for malignant transformation due to its mutations in psoriasin, p16INK4a, and involucrin, similar to that of cSCC but at a lower level[87]. However, when the AKs and cSCC lesions do appear, they produce chemokines that increase metastatic potential.
Figure 7. This image shows the extrinsic pathway insults including infection, stress, UV radiation, and carcinogens while the intrinsic pathway is due to oncogene activation[86]. UV: ultraviolet radiation; JAK: Janus kinase
Chemically induced mutagenesis
Cell growth and apoptosis
Chemicals, including arsenic and pharmacologic agents, may be implicated in the development of cSCCs. Chronic exposure to high concentrations of arsenic in food and drinking water is a common cause of cSCC, especially cSCC on the palms. Arsenic levels in drinking water as low as 300 μg/L have been found to cause cSCC[88]. Levels between 200-1000 μg/L have been identified in 2013 in the ground water in multiple communities in Massachusetts[89]. There are many medications and vaccinations that include the use of arsenic as adjuvants, active and inactive ingredients depending on the medication or vaccine; however, the amount within these pharmacologic agents is not enough to evoke malignant transformation of keratinocytes. Nonetheless, there is a need for further research on the chronic exposure of small amounts of arsenic and possible malignancies. Arsenic-exposed hyperkeratotic epithelial cells express elevated levels of keratin-1, keratin-10, involucrin, and loricrin, biochemical mediators important to the proliferation of keratinocytes[88]. These lesions can further progress to become Bowen’s disease and later cSCC with full atypia. Arsenic has also been found to change the functionality of transcription factors and transcriptional co-activators that affect cell growth and the stromal environment in which keratinocytes reside. Genetic pathways of UV-induced cSCCs commonly express TP53, HRAS, or other tumor suppressor gene mutations leading to evasion of apoptosis. However, when analyzing the signaling pathway modulation caused by arsenic, many of the genes mutated are directly involved with transcription. Nrf-2, a transcription factor important to the homeostasis of redox reactions within keratinocytes, keeps inflammation and oxidative stress at bay. Gene suppression of Nrf-2 in chronically exposed arsenic human epithelial cell lines produces arsenic-induced malignant conversion of keratinocytes[88]. Additionally, the transcription co-activator, Yap, effects keratinocytes leading to malignant transformation. Yap is only activated via phosphorylation by Phospho-LATS kinase in wild-type keratinocytes. With a high level of chronic arsenic exposure, Yap is translocated to the nucleus of keratinocytes causing increased proliferation and the eventual appearance of dysplasia and malignancy[90].
BRAF inhibitors can also affect the appearance of cSCCs. During treatment with BRAF inhibitors, cSCCs can spontaneously appear. There are many case reports showing that BRAF inhibitor, Sorafenib, inhibits PI3K, MAP kinase, and NFκB, which can reduce the release of cytokines from Langerhans cells, decreasing T lymphocyte response to new cSCCs[91]. As a result, the presence of IAKs or small cSCCs on patients taking BRAF inhibitors has the potential to cause rapid growth of cSCCs to become lacSCC due to the lack of immune response against these lesions.
Supporting stroma and vascularization
Invasion of the stroma that supports keratinocytes is a process that maintains and even evokes aggressive malignant behavior in cSCC. Arsenic and UV radiation affect keratinocyte stroma similarly. Chronic arsenic exposure leads to hypersecretion of MMPs, which degrade type IV collagen in the basement membrane. This promotes the diapedesis of immune cells and the release of inflammatory markers and chemokines, causing further cell growth and increased malignant potential. In addition to arsenic, UV also affects the stroma of underlying keratinocytes. UV radiation leads to genetic changes within the cell affecting dermal fibroblasts and the underlying basement membranes. The structure of the microenvironment, including the epidermal basement membrane and dermal extracellular matrix, can be altered by precancerous lesions. These precancerous lesions cause a release of chemokines, leading to an invasion of inflammatory cells and encourage the growth of cSCCs. These AKs and cSCCs have been found to have increases in MMPs, disintegrin-like metalloproteinases domain (ADAMs), tissue inhibitors of metalloproteinases, and other extracellular matrix degrading enzymes. Furthermore, tumor cells change the molecular function of cell-cell adhesion and prevent the integrins of damaged basement membrane from binding to extracellular matrix and binding to tumor cells. Tumor cells upregulate complement factor H and factor H-like protein 1, leading to further insult and invasion. The upregulation of these inflammatory signals by tumor cells directly affects the microenvironments of keratinocytes, causing further insult to the stroma[92].
Trauma and chronic inflammatory conditions
Chronic inflammation and trauma can change the epigenetics of the composition of the underlying stroma on which the epithelial cells lie, allowing these cells to have greater metastatic potential. The upregulation of STAT3, p63, FGFR2, and other genes coding for and upregulating chemokines can be the change necessary for conferring invasive potential[92]. The chemokine SDF-1 and its endothelial cell receptor C-X-C chemokine receptor type 4 (CXCR4), both involved in angiogenesis, are upregulated in chronic inflammatory states. Once premalignant lesions are formed, SDF-1 and CXCR4 may affect chemotaxis, upregulation of metalloproteins, and activation of stromal fibroblasts, leading to loss of collagen types in the basement membrane[93]. Marjolijn ulcers (seen in burn patients), chronic decubitus ulcers, and diabetic ulcers can all undergo processes that lead to stromal changes and subsequent keratinocyte differentiation. The signaling pathways associated with chronic inflammatory ulcer transformation can appear from a spontaneously new pathway that is also associated with the epithelial cell growth. One signaling pathway is linked with inhibiting the cdk, PDGF, and SHH pathway modulations. However, an additional pathway deals with the suppression of the proapoptotic WNT/β catenin pathway. Both pathways downregulate the extracellular matrix genes and upregulate MMP gene activation, affecting many of the same pathways affected by UV radiation. Nevertheless, Marjolijn ulcers trigger a fibrotic change in the basement membrane causing a transformation in the function of adhesion molecules between keratinocytes[94]. Marjolijn ulcers display decreased expression of IL-18, suggesting that overexpression and utilization of the immune system in chronic inflammation can eventually cause suppression of the immune response towards infections and cancerous cells[83]. This information can guide further research into chronic inflammatory conditions and trauma causing cutaneous malignancies.
Not all cSCCs are preceded by precancerous lesions. Some cSCCs can appear de novo. This is especially true in the cSCCs developed by transplant patients. Transplant patients often have weakened immune systems due to both immunosuppressive therapy and the underlying conditions that necessitated the transplant. Post-transplant patients with cSCC have been found to have de novo T lymphocyte mutations in ZNF577 coding for zinc finger proteins and FLOT gene coding for T-cell migration, dampening the immune response against tumor cells and subsequent overgrowth[95]. These types of genetic mutation pathways that lead to cSCC can be investigated as innovative treatment approaches for patients with more advanced forms of cSCC.
The tumorigenesis of cSCC is complex. UV exposure, exposure to carcinogenic substances, alteration of the stromal environment of keratinocytes, and chronic inflammatory states may all be implicated in the development of a tumor, either independently or in combination. There are multiple possible genetic drivers capable of fostering the development of cSCC. Each of these drivers may influence the development of a malignancy by impacting one of the four hallmark pathophysiological processes common to all cancers. Depending on the provoking carcinogenesis, signaling mutations of the provoking carcinogenesis, and crosstalk between the signaling of the hallmark processes, cSCC may have various aggressive potentials and require various biogenic signaling-related therapies. This is a complex multi-factorial individualized process overlapping signaling mutations that can be individually addressed therapeutically for cSCC. It is important to note that all tumor cells are not created equally. In fact, the processes, mutations, and pathways discussed above can occur at once within one tumor between multiple cells. GEP testing by Castle Biosciences, Phoenix AZ, is being developed for SCC to assess gene upregulation and downregulation within a given tumor for high and low risk patterns. This test will help determine metastasis risk among high risk cSCC patients. Thus, the GEP test, when available, should help improve prognosis projections for thus tumors labelled as poor prognosis cSCC[96].
Genetics of verrucous carcinoma
Verrucous carcinoma (VC), first described by Dr. Lauren Ackerman in 1948, is a rare variant of SCC, usually in the oral mucosa. Risk factors for VC include HPV infection, smoking, chronic inflammation, and repeated trauma. While the tumor generally has a good survival prognosis, it can be locally aggressive and may recur after treatment. The mortality with VC is more often due to local invasion rather than metastases[97]. VC usually presents as a warty, exophytic, non-ulcerating lesion with a red/white surface, making it difficult to clinically distinguish from other similar appearing dermatologic conditions. This ambiguity leads to a lack of objective criteria for diagnosis, as well as challenges in establishing a definitive method of treatment.
Malignant cell growth
While a definitive association has not yet been described, HPV infection may be a significant contributor to VC. Plantar VC was shown to be associated with HPV type 16, while VC of the scalp was associated with HPV type 33[98,99]. E6 and E7 oncoprotein production by high-risk strains of HPV cause p53 and retinoblastoma (RB) tumor suppressor inactivation, leading to cell cycle disruption and contributing to the progression of VC[100]. The E6 protein forms a trimeric complex with p53, a tumor suppressor, and E6-AP, a cellular ubiquitination enzyme [Figure 8]. This complex interferes with the functions of p53, leading to uncontrolled cell growth. The RB protein normally binds to transcription factors of the E2F family, which allows it to suppress replication enzyme genes like origin recognition complex 1, mini-chromosome maintenance proteins, and cell division cycle 6[101]. The E7 protein inhibits this interaction between E2F and RB, allowing for uninhibited cell division and replication[102].
Figure 8. The pathway of human papilloma virus (HPV). This figure describes the effects of HPV on cells involving the E7 and E7 proteins
Chronic inflammation is another hypothesized cause of cutaneous VC. VC has arisen in cases of decubitus ulcers, hidradenitis suppurativa, epidermolysis bullosa, as well as chronic bladder inflammation by schistosomiasis[103,104]. Chronic inflammation causes oxidative stress due to the release of cytokines, prostaglandins, and TNF. This leads to genetic and epigenetic modifications including the inhibition of DNA repair, modification of transcription, prevention of apoptosis, and stimulation of angiogenesis. Inflammatory cells such as macrophages and T lymphocytes may express migration inhibitory factor (MIF), which inhibits p53 transcriptional activity, leading to a loss of cell cycle regulation. The Cys81 residue of MIF is thought to play a critical role in this association between MIF and p53, leading to the inhibition of p53 mediated gene activation and apoptosis[105]. The loss of p53 contributes to a lack of response to DNA damage, which increases the likelihood of carcinogenesis.
Repeated mechanical irritation also leads to DNA damage and resultant carcinogenesis[103]. Specifically, in the case of epidermolysis bullosa, VC was thought to arise due to a mutation in the keratin 5 gene (KRT5). In keratinocytes, KRT5 and KRT14 normally copolymerize to form intermediate filaments which help anchor the epidermis[106]. Mutations of helix boundary motifs of KRT5 are thought to lead to the most severe phenotype of epidermolysis bullosa[107]. These mutations lead to fragile keratinocytes with unstable cell-to-cell junctions. This instability causes keratinocyte-mediated inflammation, which may contribute to the pathogenesis of VC[104].
Oral verrucous carcinoma (OVC) is more commonly seen than the cutaneous subtype. The PI3K/AKT/mTOR pathway was shown to be upregulated in OVC, suggesting a role for this pathway in the progression of OVC[59]. In this pathway, PI3K phosphorylates AKT, which confines AKT to the plasma membrane [Figure 2]. This is followed by many downstream effects of AKT, one of which is the activation of mTOR. mTOR is a serine/threonine kinase expressed in human cells, and its action is executed by two complexes, TORC1 and TORC2. TORC1 leads to the downstream activation of 4EBP1 and P70S6K, which are involved in the translation of mRNA into proteins for cell growth[108]. This pathway leads to increased proliferation of tumor cells, as well as resistance to treatment. The PI3K/Akt/mTOR pathway is also upregulated in oral SCC.
Furthermore, the downregulation of miRNAs contributes to the pathogenesis of OVC. Specifically, miR-195 is found to be significantly downregulated in OVC[109]. miR-195 serves as either an oncogene or a tumor suppressor, and is involved in tumor proliferation, metastasis, apoptosis, and angiogenesis. miR-195 exerts its effects through various target genes, one of which is cdk6. cdk6 is activated in the G1 phase of the cell-cycle and plays a role in the development of various cancers; cdk6 is found to be significantly upregulated in OVC[110]. This negative correlation between the expression of cdk6 and miR-195 in OVC serves as confirmation of the interaction between the two. However, the specific role of miR-195 and cdk6 in OVC pathogenesis is an area requiring further investigation.
Apoptosis evasion
The diagnostic value of expression of the apoptosis-related regulatory proteins p16, p53, p21, and RBGP (retinoblastoma gene product) in cases of oral, penile, and cutaneous VC has been studied. The variation in expression of these proteins may be helpful in differentiating VC from classic SCC[111].
P16 is a cyclin D-dependent kinase inhibitor that is inactivated in a variety of cancers. This protein blocks the progression of the cell from the G1 to S phase[112]. Interestingly, p16 additionally plays a role in the regulation of UV-induced apoptosis of cells[113]. In cases of VC, some level of p16 expression is found in 79% of cases[111]. p21, a mediator of p53 function, is upregulated by p53 in response to DNA damage. Similar to the actions of p16, p21 upregulation leads to the inhibition of cdk2 and CD4 complexes, causing cell cycle arrest in the G1 phase[111]. In 58% of VC cases, p21 expression is seen in the lower third of the epithelium, while the remaining 42% of cases show full-thickness expression[111].
While p53 mutations are one of the most frequently reported tumor suppressor mutations in human cancers, p53 expression is significantly different in VC vs. SCC. All cases of SCC showed p53 expression throughout the entire thickness of the epithelium, yet cases of VC showed p53 expression exclusively in the lower third of the epithelium[111]. Interestingly, the Ki67 protein showed a very similar pattern of expression in VC vs. SCC, with the former showing expression only in the lower third of the epithelium. The exact function of Ki67 is yet to be understood; it is thought to play a role in regulation of the G1, S, G2, and M phases of the cell cycle[114]. Both p53 and Ki67 showed a statistically significant difference in expression between VC and SCC and may play a role in differentiating these tumors.
Supporting stroma and vascularization
The differences in angiogenesis between SCC and VC have been studied using measures such as microvascular density (MVD) and endothelial proliferative index (EPI). MVD is measured by scanning CD34 immunostained sections to find areas with a high density of microvascularization. EPI is estimated using MIB-1 stained slides to locate MIB-1 positive endothelial cells. Both MVD and EPI were found to be significantly lower in VC vs. SCC; it is hypothesized that the less aggressive nature of VC may be attributable to these lower values[115].
The release of chemokines due to chronic inflammation also plays a role in angiogenesis. Members of the CXC chemokine family that are glutamate-leucine-arginine motif positive (ELR+) exert their pro-angiogenic effects by activating the CXCR2 receptor. This activation leads to increased levels of VEGF and decreased levels of thrombospondin-1, an antiangiogenic glycoprotein[116]. Stromal cell derived factor known as SCDF1 or CXCL12 plays a role in increasing endothelial VEGF expression, as well as the chemotaxis of cancer cells[117].
Modulating immunity
Chronic inflammation correlates with a lack of tumor immune surveillance. The cytokine IL-23 is upregulated in many human tumors. While IL-23 increases angiogenesis and upregulates matrix metalloproteases, one of its functions is to inhibit CD8+ T-cells, which are an essential component of tumor surveillance. Deletion of IL-23 was shown to restrict tumor growth, leading to protection against carcinogenesis[118].
The use of a TNF inhibitor, specifically etanercept, is believed to cause immune modification that may ultimately lead to the development of cutaneous VC in a patient[119]. Patients undergoing TNF-alpha inhibitor therapy may be susceptible to complex effects on the regulation of inflammation, autoimmunity, and apoptosis. These drugs have the potential to inhibit the extrinsic apoptotic pathway by modulating the activation of death receptors 4 and 5 (DR4 and DR5) [Figure 7]. DR4 and DR5 are normally activated by TRAIL, resulting in the apoptosis of cancer cells[119]. Drugs like etanercept inhibit this process, potentiating carcinogenesis. Nonetheless, a systematic review and metanalysis of patients treated with anti-TNF-α agents failed to demonstrate any increased risk of malignancy[120].
Furthermore, there is a possible effect of anti-TNF therapy on hypermethylation of the ASC/TMS1 protein. ASC/TMS1 is responsible for the activation of phagocytes through the secretion of cytokines and activation of the inflammasome[119]. ASC/TMS1 also indirectly potentiates apoptosis through its interaction with p53 and the Bcl-2-associated X protein (BAX). BAX induces apoptosis, due to the activation of caspase 8. ASC/TMS1 may be responsible for translocation of BAX to mitochondria by serving as its carrier protein[119]. Hypermethylation of ASC/TMS1 inhibits these processes, enhancing cancer development. The effect of anti-TNF drugs on ASC/TMS1 function still requires further investigation. With these known genetic signaling pathways, pharmacogenomic treatment plans can be focused on the patient and/or lesions specific genetic pathways in conjunction with the anti-TNF drugs to target all mutated signaling that can appear within patients’ varying flux states.
Genetics of basosquamous cell carcinoma
Basosquamous cell carcinoma (BSC) is a rare, aggressive skin neoplasm that has histopathological characteristics of both BCC and SCC[121]. The majority of BSC cases are found in the head and neck region, with older Caucasian males most commonly affected[122]. Clinically, BSC is indistinguishable from BCC and is commonly referred to as “metatypical basal cell carcinoma[122,123]”. Diagnosis of BSC is typically made with a biopsy of a lesion suspected of being a BCC or an SCC[124]. It should be noted that BSC is distinct from a collision tumor in that BSC is more of a “mixed tumor” consisting of BCC with areas of SCC differentiation[125], whereas a collision tumor has distinct BCC and SCC entities that are present in close proximity[126]. This can be seen histologically by the presence of a transition zone of atypia in BSC, an uncommon feature for a collision tumor[125]. Compared to BCC, BSC has a higher tendency for local recurrence and a higher propensity for lymph node and distant metastases[121,127]. BSC comprises approximately 2% of all skin cancers with a metastasis rate of about 7%[128] compared with a metastasis rate of BCC of 0.0028%-0.55%[127,129] and of SCC of 2%-5%[130]. The BSC recurrence rate is 12%-51% after standard surgical excision[128]. This contrasts with post-standard surgical excision recurrence rates of BCC and SCC at 5%-14%[131] and ~8%,[132,133] respectively. BSC has an approximately 4% recurrence rate following Mohs micrographic surgery compared with recurrence rates post-Mohs micrographic surgery (MMS) of BCC (1%-2%)[131] and SCC (3%), respectively, although there was some variability in the literature[128,131,134]. Interestingly, the recurrence rates of BSC post-MMS were reported to be about 4% regardless of site (head and neck, trunk, lower limb, or upper limb). In BSC larger than 2 cm, the rate of recurrence is believed to be slightly increased[128]; however, these results were not statistically significant, likely due to small sample size[134]. Thus, MMS is currently the preferred treatment for BSC independent of body location.
Malignant cell growth
BCC genetic drivers’ role in BSC
BSCs are frequently associated with SHH pathway mutations, implicating SHH deregulation as the primary driver in BSC and providing evidence that BSCs shares similar cancer drivers to BCC[135]. Loss of function in PTCH1 and gain of function in G-protein-coupled receptor SMO in the BCC pathway are the most common mutations that cause SHH deregulation in the BCC pathway. Similarly, 45% of BSCs were shown to have deleterious mutations in PTCH1 compared to 44% of BCCs and 10% of SCCs. About 5% of BSCs contain SMO oncogenic mutations compared with 25% of BCCs and 0% of SCCs[135]. BSCs also contained all the other known BCC cancer drivers, MYCN, PP6C, GRIN2A, CSMD3, DCC, PREX2, APC, and ARID1A at a frequency statistically similar to that of BCCs[135].
SCC genetic drivers’ role in BSC
BSCs lack the classical SCC driver mutations, NOTCH1/2, HRAS/KRAS, and CDK2NA, at a frequency expressed by SCCs. Instead, BSC contain SCC driver mutations at a significantly lower rate that is closer to that seen in BCCs. The presence of classical BCC cancer drivers in BSCs and the lack of classical SCC driver mutations suggests that BSCs have a mutational landscape similar to that of BCC, and that BSC cancer drivers likely arise through the deregulation of SHH signaling[135].
Genetics of cells modulating between BCC to SCC cells
It is believed that BSC originally derive from BCC due to their similar mutational landscape; they subsequently acquire mutations that lead to squamatization[121]. Of the 20 cancer genes identified in BSC, ARID1A is mutated in 45% of BSCs compared to 19% of BCC and 19% of SCC[125]. ARID1A normally plays a role in the differentiation of several cancer types because it encodes a component of the chromatin remodeling complex, SWI/SNF. SWI/SNF plays an important role in repairing damaged DNA, so ARID1A impairment can cause defects in DNA repair. Disruption of ARID1A reduces the restrictive nature of chromatin remodeling in terminally differentiated cells, imparting a plasticity that increases cell survival, regeneration, and proliferation. Under selective pressure, such as a SMO inhibitor treatment, ARID1A mutations allow keratinocytes to undergo squamatization, promoting de novo SCC development from BCC[125]. Further research could be done to see if mutations within other components of chromatin remodeling could confer plasticity in BSC.
Additionally, switching from the SHH pathway to the RAS/MAPK pathway is a form of crosstalk between the pathways and another way that BCC can avoid selective pressures such as SMO inhibitors, leading to squamatization. As discussed in the SCC section, an upregulated RAS/MAPK pathway can lead to SCC. Pathway switching is believed to be driven by a loss of primary cilia, a microtubule-based signaling organelle that is essential for high SHH pathway signaling, promoting RAS/MAPK signaling and subsequent squamatization as seen in Figure 9. Furthermore, Gli1 is active in the nucleus and is typically elevated in BCC. Stained BSC shows that basaloid keratinocytes demonstrate high Gli1 and low MAPK staining which is similar to that of BCC. Squamatized keratinocytes in BSC demonstrate low Gli1 and high MAPK staining which is similar to that of SCC. Interestingly, the transition zone shows a moderate MAPK expression and high Gli1 expression, indicating that RAS-MAPK pathway activation drive squamatization in BSC with subsequent loss of Gli1 expression as a secondary event. This discovery indicates the activation of the RAS-MAPK pathway and subsequent reduction in SHH signaling as a potential modulator of tumor plasticity in BSC[136]. Overall, tumor fate in BSC is believed to be dictated by a balance between SHH and RAS/MAPK signaling[125].
Figure 9. Genetic lineage of BSC. A: activating SHH pathway mutations initially drive formation of BCCs. Acquisition of de novo ARID1A mutations or other chromatin remodeling mutations under pharmacological SMO inhibition drive cellular plasticity, pushing basal cells to undergo squamatization and leading to BSC formation; B: within the BCC zone, high levels of SHH signaling, low RAS/MAPK pathway activity, and high levels of ciliation drive tumor growth. Within the SCC zone, RAS/MAPK signaling increases with a concomitant reduction in SHH pathway activity and ciliation. Within the transition zone, ce3lls begin to show higher levels of RAS/MAPK pathway activity while maintaining SHH signaling. The levels of ciliation are unknown. SHH: sonic hedgehog; BCC: basal cell carcinoma; SMO: Smoothened; BSC: basosquamous cell carcinoma
Calcium flux is a potential modulator of the balance between SHH and RAS/MAPK signaling in BSC. The degree of extracellular calcium is a regulator of the SHH pathway. In rat gastric mucosal cells, SHH is unable to activate extracellular signal-related kinases in calcium-free conditions. Conversely, cells in a calcium-rich media show increased intracellular calcium levels, a marker for SHH pathway activation[137]. Altogether, it is possible that calcium flux shifts the balance between SHH and RAS/MAPK signaling in BSC, contributing to tumor plasticity[138]. More research is needed to establish a causal relationship between calcium flux and the balance of SHH and RAS/MAPK. Future studies could be done to identify additional modulators and to understand the interplay between existing modulators in conferring tumor plasticity.
Though it is known that BSC appear from BCC, it is unknown if these cutaneous lesions can also appear from a SCC. However, due to the consistent state of flux that the body undergoes and crosstalk between these genetic pathways, there is a possibility that SCCs could become BCCs and BCCs can become SCCs. There is a need for further research finding data to address this issue.
Prevention of apoptosis
Strong expression of FasL in both BCC and SCC is believed to allow the tumor to avoid apoptosis. FasL mediates apoptosis in cells that express Fas receptor and is a member of the tumor necrosis family. FasL on tumor cells can bind to the Fas receptor on T-cells, macrophages, and natural killer cells which make these effector cells unable to infiltrate the tumor nodules and trigger the extrinsic apoptotic pathway[139]. Although there have not been studies confirming that strong expression of FasL is an important mode for how BSCs prevent apoptosis, BSCs’ genetic similarity to BCC and histopathologic characteristics of both BCC and SCC suggest that strong FasL expression plays a role in allowing BSC to avoid apoptosis. BSC that do not have strong expression of FasL may not be able to avoid apoptosis or may avoid apoptosis through other means.
Additionally, bcl-2 promotes cell survival without stimulating cell proliferation. Bcl-2 is associated with the intrinsic apoptotic pathway, triggered by intracellular stress[140]. Studies indicate that bcl-2 positive tumors have a better prognosis and slower course of progression. In BSC, immunohistochemical studies for bcl-2 yield a positive result in the BCC regions but a negative result in both the transition zone and SCC region[141]. Studies show only low-grade bcl-2 positivity in the BCC regions of BSC, similar to that of aggressive BCC. In contrast, non-aggressive BCC such as nodular and superficial BCC have high grade bcl-2 positivity. This suggests that BSC may downregulate bcl-2 in the BCC regions, stimulating uncontrolled cell proliferation and contributing to its more aggressive behavior compared to non-aggressive BCC[141].
Crosstalk
Crosstalk between various signaling pathways can be a determining factor as to why some patients respond to cancer treatments and others fail treatments. In fact, the BSC pathways and hair follicle pathways appear to have significant crosstalk; both of these pathways are dependent on SHH, PTCH1, SMO, and Gli[142]. In normal hair follicle development, PTCH1 acts as a receptor for SHH, and their interaction triggers SMO which modulates a signaling cascade. Activation of the SMO cascade results in subsequent translocation of Gli to the nucleus. Gli is a key modulator of the SHH pathway and causes upregulation of several downstream target genes that modulate hair follicle development[143]. Injury to skin enlists hair follicle stem cells in the healing process, namely through the SHH pathway. However, if cells that carry a mutation in SMO are recruited to the injury site, this mutation will trigger unregulated downstream SHH pathway signaling which is implicated in BCC and BSC[144]. This shared SHH pathway involvement suggests that the mechanisms of tumor formation and hair follicle proliferation are interlinked and essential for the normal development of hair follicles with dysregulation or mutation in this pathway leading to cancer[143]. It is possible that BSC originates as BCC from pluripotent basal cells and initiates in a similar manner as BCC, typically from hair follicle stem cells in the hair follicle bulge[145], as seen in the previous BCC section. Through modulating pathways conferring plasticity as described above, squamatization occurs to form BSC.
Vismodegib is a SHH pathway inhibitor that is used for the treatment of BCC. There have been many cases of SCC developing from BCC following treatment with Vismodegib. The appearance of SCC may indicate a de novo SCC adjacent to the BCC which is proliferating independently, an SCC that develops as a result of stem cell differentiation during SHH inhibition, or an SCC that was present as part of a metatypical BCC such as BSC that has decreased SHH signaling due to SHH inhibition and subsequently increased RAS/MAPK signaling[146]. There fails to be many reported cases of SCC transforming to BCC following use of Vismodegib. Thus, it may be difficult to determine if SHH inhibitors could play a role in the desquamatization of cSCC to BCC. Nevertheless, because BSC has a high tendency for recurrence and has a high rate of metastasis, Moh’s micrographic surgery is currently the preferred treatment for BSC[128].
Supporting stroma and vascularization
When comparing supporting stroma of BCC and BSC, there is a significant difference between the stroma of BSC, and high risk (HR) BCCs (micronodular and/or infiltrative) compared to that of low risk (LR) BCCs (nodular and/or superficial). LR BCC group lesions have fibromyxoid stroma without desmoplastic stroma, whereas 80% of the HR BCC and 61.8% of the BSC group lesions have desmoplastic stroma[147].
The stromal differences in LR and HR BCCs and BSC further suggests variety between the tumor types. Fibromyxoid stroma is most often observed in lesions with mild inflammation. However, desmoplastic stroma is associated with dense inflammation which attributes to stroma-derived factors that recruit additional inflammatory cells or inflammatory cell secreting factors that change stromal characteristics[147]. Inflammatory cell/secreting factor recruitment of collagenolytic enzymes that cause destruction of the surrounding collagen and matrix metalloproteinases that cause proteolysis of extracellular matrix components are implicated in SCC and likely BSC to cause desmoplastic stroma, facilitating tumor growth and metastasis[148]. Further research needs to be done to identify the types and functions of immune cells in peritumoral inflammation in order to understand the interactions between peritumoral inflammation and stroma specifically in BSC. Additionally, activated fibroblasts and inflammatory cells of peritumoral stroma secrete extracellular matrix proteins and growth factors in a paracrine fashion which can change the expression of genes affecting angiogenesis, tumor growth, and metastasis. Though the precise mechanisms underlying the complex interactions between the stroma and tumor in BSC is unknown, the crosstalk communication between various genetic signaling pathways indicates the flux state amongst signaling pathways and keratinocyte homeostasis.
Modulating immune response
Dense peritumoral/perivascular inflammation in BSC is a marker of host immune response and may be associated with recruitment of protumor immune cells. For LR BCC, 83.3% of the lesions exhibit only mild peritumoral inflammation whereas of the HR BCC, 76% of the lesions exhibit dense peritumoral inflammation. Of the BSCs, 51.4% demonstrate dense peritumoral inflammation and 37.1% demonstrate moderate peritumoral inflammation, frequencies that most closely approximated those of the HR BCC group[147]. Similarly, perivascular inflammation in adjacent dermis was observed in 91.4% of BSC group lesions, 97.5% of HR BCC group lesions, and 55% of LR BSC group lesions. Only the BSC and HR BCC group lesions were found to have dense perivascular inflammation[147].
The host immune response between BSC and HR BCC shows some similarities based on peritumoral and perivascular inflammation density, both in contrast to LR BCC. Firstly, differences in inflammation density between BSC and LR BCC may be attributed to an increased number of regulatory T-cells in BSC that suppress anti-tumor T-cell response and are associated with a worse prognosis compared to LR BCC that do not have increased regulatory T-cells[147]. Secondly, immature dendritic cells (IDCs) are involved in attenuating the immune response in BCC and perhaps also in BSC. Attenuation of the immune response occurs through the induction of peripheral T-cell tolerance by IDCs and through regulatory DC secretion of IL-10 that suppresses T-cell proliferation. IDCs are also believed to contribute directly to tumor proliferation through unknown mechanisms. Lastly, an increase in expression of Th2 cytokines, specifically IL-4 and IL-10 contribute to an immunosuppressive tumor environment that favors proliferation in BCC and perhaps also BSC[149]. It should be noted that the tumor permissive mechanisms described above likely occur concurrently with immune responses directed towards tumor eradication. Anti-tumor immune responses include: CD8+ T-cells for a specific adaptive antitumor response, IL-23 for enhanced proliferation of memory T-cells, IL-12 for activation of mature DCs and Th1 antitumor immunity[149]. Overall, BSC modulation of immune response appears to occur dynamically with a protumor, attenuated immune state only partially compensated for by the host antitumor response. This dynamic state shows myriad fluctuations and crosstalk signaling between genetic signaling pathways, which can further personalized BSC treatment for advanced or metastatic BSCs.
Genetics of melanoma
Although NMSC are the most common type of skin cancer, cutaneous melanoma is the most aggressive, accounting for greater than 80% of skin cancer deaths largely due to its susceptibility to metastasize to other organs[150]. Close to 200,000 cases of melanoma will be diagnosed in the United States in 2020 with a rate of one American dying from melanoma every hour[151]. There are 4 main types of melanoma: superficial spreading melanoma (SSM), nodular melanoma (NM), lentigo maligna melanoma (LMM), and acral lentiginous melanoma (ALM)[152]. Cutaneous SSMs account for 70% of melanomas, while NMs account for 15%-30% of melanomas. Both SSM and NM subtypes are often associated with the BRAF V600E mutation[152]. ALM (2%-10%) and LMM (5%) are rarer types that are both associated with mutations in the C-kit gene[152].
The transformation of melanocytes into melanoma cells and further progression to metastasis involves the complex interaction of signaling pathways with multiple environmental, genetic, and host factors. The primary contributor is DNA damage from UV light exposure, although genetic disorders like xeroderma pigmentosum and familial history of melanoma can strongly increase a person’s risk[153]. Occupational exposure to ionizing radiation among radiologic technologists has been shown to increase risk to developing skin cancer and melanoma[154]. A recent literature review suggests airline pilots and cabin crew may have twice the risk of melanoma compared to the general population and increased melanoma mortality among pilots from possible cumulative cosmic radiation[155]. However, much of the relevant evidence is considered out-of-date as it reflects environment and behaviors in the late twentieth century and is incongruent with modern day standards. Immunosuppressed populations like organ-transplant recipients have an increased risk of many malignant cancers, with skin cancer being the most common[156]. Understanding the mechanisms in various signaling pathways can provide insight into providing personalized and effective treatment. This amalgamation of genetic crosstalk modulates the transformation process and involves the hallmarks of cancers: (1) cell growth; (2) prevention of apoptosis; (3) supporting stroma and vascularization; and (4) modulating immune response.
Cell growth
UV-induced damage
Within hours, UVA causes immediate and lasting hyperpigmentation (tanning) due to induction of oxidative stress in melanocytes. UVA damages the extracellular matrix and induces an immune response facilitating invasion and metastasis of skin cancer cells[113]. In contrast, UVB directly causes skin cancer genesis and induces a slower delayed tanning through a nascent melanin synthesis pathway and melanocortin receptor-1 (MC1R) signaling[3]. MC1R activates the DNA damage response causing the formation of cyclobutene pyrimidine dimers and 6-4 photoproducts that distort the DNA helix and when unrepaired lead to mutations[157]. In addition, UVB triggers an inflammatory response by recruiting neutrophils and macrophages and promotion of angiogenesis contributing to melanoma cell survival and metastasis[3].
CDKN2A/p16 deletion
The CDKN2A/p16 protein is a crucial cell cycle gatekeeper at the G1-S checkpoint and its location encodes for tumor suppressors p16INK4a and p14ARF[158]. As mentioned in the cSCC section, P16INK4a inhibits cdk4 and cdk6 [Figure 10], activating RB protein and preventing cell cycle progression into S from G1 phase[159,160]. On the other hand, p14ARF positively regulates p53 [Figure 10] by inhibiting negative regulator MDM2[161,162]. Deletions in the CDKN2A locus was found in 50% of all melanomas and had high penetrance in familial melanoma[153,163]. The inactivation of CDKN2A and p53 inactivation leads to uncontrolled cell proliferation of melanocytes.
Figure 10. Significant signaling pathways in melanoma. Schematic of major pathways associated with cell survival, differentiation, and proliferation. Arrows represent active signals and red lines represent inhibitory signals. mTOR: mammalian target of rapamycin; MITF: microphthalmia-associated transcription factor
Apoptosis evasion
BRAF in RAS/RAF/MAPK/ERK pathway
The most prevalent and highly studied oncogenic melanoma mutation is BRAF with the most common mutation substituting glutamic acid for valine (V600E)[164]. An estimated 40%-50% of mutated melanomas are of BRAF V600E mutation[165]. In the MAPK pathway [Figure 10], the presence of the oncogenic BRAF V600E mutation drives constitutive phosphorylation of MEK1 and 2, activating ERK 1 and 2 and reprograming cellular metabolism to sustain cell survival and growth[166]. In major melanoma subtypes of NRAS, BRAF, and c-kit pathways, proteins expressed in the MAPK signaling pathway has been found suggesting crosstalk between pathways[167]. In addition, several mutations of key upstream proteins of the aforementioned MAPK cascade includes NRAS, KIT, GNAQ, and GNA11[168]. In over 80% of uveal melanomas, GNAQ or GNA11 is activated using the MAPK pathway[169].
PI3K-AKT pathway
The neuroblastoma RAS viral oncogene (NRAS) is mutated in approximately 30% of melanomas[168]. Ras binds to PI3K activating secondary messenger PIP3 and binds to serine threonine kinase AKT[170]. An important downstream effector of PI3K-AKT is mTOR [Figure 10], which functions to initiate a cascade that inhibits autophagy[160,171]. PTEN is an antagonist of PI3K-mediated signaling. Loss of functional tumor suppressor gene PTEN increases AKT phosphorylation leading to decreased apoptosis and increased melanoma survivability[165]. PTEN mutation was also found in nearly 44% of BRAF mutated melanomas in contrast to 4% of NRAS mutated melanomas[168]. These mutational patterns suggest that BRAF and NRAS mutations appear to be mutually exclusive and distinct oncogenic drivers[170].
c-KIT inhibition
c-KIT encodes for transmembrane tyrosine kinase KIT which binds to stem cell factor and activates cellular proliferation pathways including RAS-ERK and PI3K/AKT [Figure 10][162,172]. KIT mutations were more frequently in mucosal, acral, and chronically sun-damaged skin melanoma than in non-chronic sun-damaged skin melanoma in the United States[173]. Of note, KIT mutations are believed to be more frequent in non-white populations and reported in higher mutation rates in East Asian patients[174,175].
Supporting stroma and vascularization
HIF-1α/VEGF
Tumors require vascularization to grow, metastasize, and invade. Angiogenesis correlates with the progression of melanoma tumor growth by supplying oxygen and nutrients and providing an opportunistic route to spread into blood circulation. VEGF is an important angiogenic factor and leads to secretion of MMPs degrading extracellular matrix and allowing for openings for cells to invade neighboring tissues[174]. HIF-1α initiates VEGF-A expression and key to downregulating mitochondrial energy metabolism by switching to glycolysis[163]. This Warburg effect of lactic acidosis contributes to the acidification of the tumor microenvironment triggering metabolic reprogramming in melanoma cells [Figure 11] with increased pro-angiogenic VEGF and altered immunosuppression[167].
Figure 11. Multifactorial reprogramming to acidification of melanoma tumor microenvironment. The pH and oxygen microenvironment work together with genetic and metabolic pathways to promote melanoma proliferation and metastasis progression[176]. HIF-1α: hypoxia-inducible factor-1 alpha; mTOR: mammalian target of rapamycin
Melanocyte-lineage
Microphthalmia-associated transcription factor
The microphthalmia-associated transcription factor (MITF) gene is a key regulator for the development and differentiation of melanocytes. Melanoma cell phenotype switching is characterized in the ERK/MAP-kinase pathway [Figure 10] targeting MITF phosphorylation in either transient or sustained activation[177]. Sustained ERK phosphorylation inhibits MITF keeping its levels low leading to proliferation. High MITF levels along with transient ERK phosphorylation triggers melanocyte differentiation[178]. cAMP signaling activates HIF-1α and increases MITF levels[179]. HIF-1α and VEGF promotion triggers microenvironment changes that promote melanoma progression [Figure 11]. Cellular crosstalk in signaling pathways also plays a driving role. MITF activates anti-apoptotic gene BCL2, promoting melanocyte survival[180]. Cell cycle inhibitors p21 has been implicated along with p16INK4A and p14ARF [Figure 10] in the senescence of melanocytes[181]. As the fate regulator of the melanocyte lineage, MITF activity fluctuates through modification of signaling pathways and microenvironmental dynamics.
Modulating immune response
Melanoma utilizes co-inhibitory pathways to avoid surveillance by the immune system and its ability to respond and possibly eradicate the tumor cells. Two well-characterized co-inhibitory pathways involve the cytotoxic T-lymphocyte associated antigen 4 (CTLA-4) and programmed death-1 (PD-1) T-cell co-receptor[182]. CTLA-4 outcompetes CD28 for B7 binding on antigen presenting cells, limiting IL-2 production and lowered survivability of the T-cell[183]. PD-1 is induced later during T-cell activation and ligation with PDL1 and PDL2 promote tumor growth[184]. Immunotherapy for melanoma blocks PD-1 and CTLA-4 checkpoints and elicits a therapeutic response[183]. The role of mechanical trauma in the development of melanoma remains unclear. One retrospective analysis on acral melanoma accounting for the majority of melanoma in the Chinese population provided epidemiological evidence for potential association between trauma and acral melanoma[185]. For dark-skinned and East-Asian populations, patients have reported direct trauma related to subungual melanoma[186]. However, there is not enough literature to support an established correlation.
Future directions
Many patients with good prognosis indicated by early stage through traditional clinicopathologic histology staging methods still decline from metastases[187]. Recent advances in personalized medicine could be instrumental in capturing a more accurate and detailed micro-staging of melanoma through next-generation sequencing and genetic profiling. Validation studies of the 31-GEP, which categorizes risk as low or high by class, has indicated the 31-GEP test as an accurate predictor of metastasis of cutaneous melanoma[188-190]. Systematic meta-analysis demonstrated the 31-GEP test consistently identifies melanoma patients at increased risk of metastasis independent of clinicopathologic factors and improves on current staging[191]. The identification of early stage low-risk patients vs. late stage high-risk stage patients would significantly help in determining life-saving adjuvant therapies or avoiding unnecessary treatments and costs. Analysis of profiles of gene expression modification induced by different signaling pathways in melanoma cells has considerable potential in understanding clinically relevant molecular mechanisms and biomarkers for non-melanoma skin cancers to create personalized treatment plans for each patient with melanoma. Unfortunately, the use of 31-GEP testing on thin melanomas is controversial. A recent article believes that the testing should not be done on people with thin melanomas if there is no evidence on change of outcome and treatment plan. Once further studies on 31-GEP testing are done, the evidence of effectiveness in thin melanomas will be apparent[192].
Summary
Recent endeavors into understanding the pathways involved in the development and metastasis of skin cancer into melanoma has led to some degree of hope in therapy against this historically difficult to treat cancer. It is clear melanoma is not homogenous, but a complex multifactorial disease characterized by crosstalk of several different pathways and elucidation of mechanisms that contribute to tumor growth and chemoresistance. Hence, understanding these complex mechanisms is crucial and will lead to more optimal treatments for each patient.
Cutaneous T-cell lymphoma
Cutaneous T-cell lymphoma (CTCL) is a type of T-cell mediated dermatologic neoplasm which falls under non-Hodgkin’s lymphoma[193]. Mycosis fungoides (MF) and Sézary syndrome (SS), the most frequently diagnosed forms of CTCL, have become some of the most researched forms of non-melanoma skin cancers. CTCLs are often misdiagnosed as benign skin conditions, such as atopic dermatitis or psoriasis[194]. Understanding the various signaling pathways of CTCL is conducive to forming proper treatment plans and prognoses. The goal of this section is to discuss the pathways in which CTCL arises and the treatment options utilized for this form of non-melanoma skin cancer.
There are various theorized and studied pathways in which CTCL arises. The majority of these cases do not progress to systemic malignancy, rather, it remains a mostly cutaneous cancer. Clinical features of CTCL can be fairly variable. From erythrodermic exanthems to flesh colored papules, CTCL can present in a number of cutaneous manifestations. There are a few exogenous factors to note that have been studied to have an antigen induced CTCL reaction. A common pathway in which CTCL evades the body is via the antigen-antibody mechanism. In the antigen-antibody mechanism, T-cells are consistently exposed to specific antigens resulting in proliferation and failure to induce cellular apoptosis[195]. Not only are medications thought to be provoking factors in antigens causing T-cell proliferation, but viral, bacterial, and fungal antigens are also factors causing uncontrolled T-cell proliferation[195]. Some of these viral antigens include human T-cell lymphoma virus, Epstein-Barr virus, and herpes simplex virus. In addition, methicillin resistant Staphylococcus aureus (MRSA), Mycobacterium leprae, Chlamydia pneumoniae, and even fungal dermatophytes can be inducing factors causing CTCL[196,197]. Furthermore, there is an association between vitamin D deficiency, and even vitamin D receptor single nucleotide polymorphisms, causing deficiencies within the system. Vitamin D is important for the function of many immune cells including those of T cells. Paracrine communication between B and T lymphocytes, Cathledicin gene regulation and innate immunity all function with the use of Vitamin D. Without properly regulated innate and adaptive immunity, Staphylococcus aureus and other bacteria and viruses have a higher probability of colonizing the skin while continuously priming and activating T lymphocytes. As mentioned earlier, Staphylococcus aureus has a known association with CTCL meaning that Vitamin D and polymorphisms in the VDR can indirectly have an association with CTCL[198].
Some studies have found association between atopy and CTCL with an IgE increase found in biopsies. However, there is a need for further research in the subject of atopy and allergen-induced CTCL.
Extrinsic exposures
Various studies have determined that if the antigen producing agent is removed, then the CTCL response will eventually eliminate itself. When comparing patients with CTCL and hypertension taking hydrochlorothiazide to those with CTCL that are not taking hydrochlorothiazide, complete clearance or improvement of their CTCL cutaneous lesions has been observed when hydrochlorothiazide is withdrawn[195]. What is most important is that it is understood the different ways in which CTCL can be treated. Disruption of specific pathways via monoclonal antibody addition, as well as removing an antagonizing antigen, are just a couple examples in how this research has allowed for the elimination of this cutaneous neoplasm.
MRSA has also been found to be an insult leading to CTCL. Moreover, the treatment of MRSA can lead to improvement of CTCL. Patients with erythrodermic CTCL were discovered to have a decreased body surface area affected when being treated for a MRSA infection[196]. This finding leads to the conclusion that patients with erythrodermic CTCL have obtained a coinfection MRSA. Once treated, the cutaneous manifestations dramatically decrease, thus, it is recommended that patients with CTCL be empirically treated for a MRSA infection[196].
While many of the signaling pathways discussed are being studied to better understand CTCL progression and initiation, there are various other pathways continuing to be discovered and evaluated in association with this cutaneous neoplastic disease. Cutaneous T-cell lymphoma is a result of malignant T-lymphocytes proliferation and invasion[194].
Apoptosis evasion
Epigenetics
Many of the identified common mutations in CTCL are in genes coding for proteins that alter epigenetic factors, including AT-Rich Interaction Domain 1A (ARID1A) and DNMT3A[199]. ARID1A alters histone structure, exerting its effects through epigenetic mechanisms. Mutations in ARID1A have led to the downregulation of PTEN in other types of cancer[200]. The effect of ARID1A downregulation leads to decreased apoptosis and uncontrolled cell growth between the G1 and S phase of the cell cycle. DNMT3A is a gene that encodes for DNA methyl transferase 3α, and mutations in DNMT3A are linked to aberrant cytosine methylation and potentially the downregulation or silencing of important oncogenes[201].
One commonly researched target for CTCL is the NOTCH family of proteins. NOTCH1 activity is increased in both SS and tumor stage MF. The activity of NOTCH1 is typically upregulated through increased levels of binding ligand, Jagged1. Jagged1 ligand binds through a juxtacellular mechanism to NOTCH1. NOTCH1 is then cleaved into ICN1 by proteases. ICN1 exerts downstream effects, one of these being the expression of hairy and enhancer of split-1 (HES1) [Figure 12][202]. In tumor stage MF, the suspected upregulated ligand is Jagged1. This is thought to occur through an epigenetic pathway involving DNA hypermethylation of the miR-200c promoter region[202]. miR-200c can function as an oncogene that inhibits activation of NOTCH1 through decreasing expression of the NOTCH1 ligand Jagged1. However, hypermethylation in the promoter sequence of miR-200c decreases expression of miR-200c is seen in MF. This results in increased NOTCH1 activity through an increase in levels of Jagged1[202]. Overactivity of NOTCH1 has been linked to decreased expression of PTEN and degradation of p53 in T-cell acute lymphoblastic leukemia[203]. In addition, HES1 expression is also linked to the upregulation of the NF-κB pathway in T-cell acute lymphoblastic leukemia[204]. There is a need for more research on the cause of hypermethylation of these miRNA regions that cause downregulation.
Figure 12. Common pathways in cutaneous T-cell lymphoma. This figure describes the genetic signaling pathway of NOTCH1. NF-κB: nuclear factor-kappa B; JAK: Janus kinase
NOTCH1 is also linked to the AKT pathway through PTEN and p53 regulation by miR-122. miR-122 activates AKT[205]. TP53 mutations are found in CTCL[199]. miR-122 is primarily known for causing apoptosis within hepatocytes, yet recent studies have found that miR-122 is also found throughout human skin, especially in patients with MF. Lesioned skin has been found to express higher levels of miR-122 in patients with MF and SS compared to healthy skin. Though the normal function of miR-122 is found to induce apoptosis in hepatocytes, when upregulated, miR-122 blocks apoptosis. Cells with upregulation of miR-122 have been discovered to have phosphorylated AKT and downstream transcription factors in the pathway. As a result, AKT is then overactivated, leading to decreased apoptosis from increased TP53 inhibition. This lack of apoptosis leads to the uncontrolled cell growth of cutaneous T-cells[205].
Malignant cell growth
Mutations upregulating the Janus kinase (JAK) and signal transducer and activator of transcription proteins (STAT) are implicated in CTCL. JAK1, JAK3, STAT3, and STAT5B are mutated in certain CTCL cell lines[206]. Mutations in JAK1, JAK3, and STAT5B have also been implicated in T-cell prolymphocytic leukemia[207]. The effects of JAK/STAT signaling in CTCL are thought to be driven partially by the production of cytokines and angiogenic factors[194]. Generally, the mechanism of JAK/STAT signaling is as follows. Specific ligands or cytokines bind to the RTK, causing activation and dimerization of the receptor. This dimerization allows JAK proteins to come into close proximity to each other. JAK-JAK phosphorylation occurs, and JAK then phosphorylates STAT proteins. STAT proteins then dimerize and translocate to the nucleus, where they act as transcription factors [Figure 12][208].
Activated JAK is also known to activate PI3K, resulting in eventual activation of AKT [Figure 12][208]. Activation of AKT has also been implicated to occur in CTCL through mutations in the PI3K pathway relating to TCR-CD28[209]. Additionally, loss of heterozygosity in PTEN, the negative regulator of PI3K, have been observed in MF, and PTEN activity is shown to be altered in CTCL[194]. The PI3K pathway is explained in greater detail in the SCC section of this paper. Activated JAK is also known to activate PI3K, resulting in eventual activation of AKT [Figure 12][208]. Activation of AKT has also been implicated to occur in CTCL through mutations in the PI3K pathway relating to TCR-CD28[209].
NF-κB pathway activity has also been shown to be increased in CTCL[206]. The canonical NF-κB pathway occurs as follows. IkB kinase (IKK) acts to phosphorylate the IkB element of NF-κB-IKB. Phosphorylated IkB is ubiquitinated, which allows for the degradation of IkB by proteasomes. NF-κB is then able to enter the nucleus to act as a transcription factor[204]. One of the effects of NF-κB signaling is an upregulation of its negative regulator IkB. This serves as a negative feedback mechanism of NF-κB signaling [Figure 12][210]. The NF-κB pathway is also linked to the activation of AKT. One of the actions of AKT is the phosphorylation of IKKα, which is part of the IKK complex of the canonical NF-κB pathway [Figure 12].
The non-canonical NF-κB pathway is implicated also in CTCL. Mutations are seen in the C terminal of the gene NFκB2, which is an area encoding for autoinhibitory functions in NF-κB2. Typically, the C terminal of inactive NF-κB2 assists in suppressing the canonical NF-κB pathway. Mutations in the NFκB2 gene are, therefore, thought to contribute to activation of the canonical NF-κB pathway in CTCL[199].
The genes phospholipase C, gamma 1 (PLGC1), and caspase recruitment domain family member 11 (CARD11) are amplified in CTCL and are thought to contribute to abnormal TCR signaling[199]. PLGC1 activates the NF-κB pathway and nuclear factor of activated T-cells through diacylglycerol and inositol 1,4,5-trisphosphate (IP3), respectively[211]. CARD11 is linked to the induction of the NF-κB pathway, although the mechanism of action is not yet completely understood[212].
Other mutations in tumor suppressor genes, CDKN2A, TNF-α Induced Protein 3 (TNFAIP3), zinc finger E-box binding homeobox 1 (ZEB1) and FAS, have been identified in CTCL[199]. TNFAIP3 protein works to inactivate IKK when the NF-κB pathway is overactivated by TCR[213]. ZEB1 is a zinc finger transcription repressor protein, and the loss of ZEB1 in CTCL is linked to the overexpression of cytokines like IL-2. Mutations in ZEB1 also increases the expression of GATA3, which binds to the promoter site of the gene encoding for thymocyte selection-associated HMG box protein (TOX)[199,214]. TOX is a mediator of T-cell maturation from CD4+and CD8+ to CD4+[214]. The loss of FAS in CTCL cell lines most likely also contributes to apoptosis resistance[199].
As mentioned previously, the pathogenesis and progression of CTCL are believed to be influenced by alterations to the local cytokine milieu. These changes are thought to occur in part due to disfunction in CXCR4[215]. CXCR4 mediates downstream effects of various receptor tyrosine kinases, one of which is TCR. When TCR is activated, CXCR4 is able to ligate with TCR, triggering a process that enhances the stability of mRNA transcripts for IL-2, IL-4, and IL-10. This process occurs in the presence and in the absence of CXCL12, the sole endogenous ligand for CXCR4. Furthermore, blocking the association between CXCR4 and TCR decreases mRNA transcript stability for both TCR-dependent and TCR-independent cytokine production in SS cell lines, without altering other downstream effects of TCR[215].
Supporting stroma and vascularization
More recently, CTCL progression via angiogenesis is being studied. Angiogenesis via the VEGF pathway results in the congregation of tumor cells[216]. The stromal involvement of CTCL involves a CXCL12[216]. The stromal factor and its adjacent receptor work to create a signaling network for bone marrow stem cells, which is found in earlier disease states[216].
Summary
Though there is a need for further research on CTCL, there are many treatment options available. Historically, first line treatment for CTCL has been psoralen and UVA (PUVA) therapy[217]. When used in conjunction with interferon-α, efficacy has been shown to increase due to elevated levels of apoptosis[217]. PUVA upregulates p53 allowing increased apoptosis within the tumor cells[217]. With the addition of interferon-α, apoptosis is increasingly regulated via the JAK1 signaling pathway[217].
Brentuximab vedotin is also a potential treatment for CTCL[218]. This monoclonal antibody has been shown to have efficacy against refractory cases of CTCL, as well as peripheral T-cell lymphoma[218]. During the studies performed using this medication, it was found that success is greatest in those CTCL cases where CD30+ positive cells are present. CD30+ falls within the TNF receptor class, which is involved in cell proliferation[218]. Thus, explaining the mechanism of this monoclonal antibody to inhibit specifically CD30+ expression[218]. Another monoclonal antibody used in the treatment of MF and SS is Alemtuzumab[219]. Thus, research of immunomodulatory treatments is on the rise for CTCL leading to personalized treatments targeted to the genetic signaling pathways, which are dependent on the person’s specific flux state.
Conclusion
Melanoma and non-melanoma skin cancer are some of the most prevalent cancers that can lead to death if not properly managed. However, knowing the genetic signaling pathways of these conditions can further help researchers and providers give more options to patients suffering from these conditions. With this information, the provoking genetic factors can be blocked with the use of pharmaco- and immunotherapy allowing for patients with treatment resistant, locally advanced and even metastatic lesions to be treated due to the different options of therapy blocking multiple signaling pathways. Though some information is known about the genetic pathways of cutaneous malignancies, there is still a demand for research that can be done to improve treatment options to decrease mortality rate and recurrence.
Initiators and promoters
There are different provoking aspects that cause cutaneous malignancies. Promoters are products or a low-level exposure a carcinogen that can begin the process of tumorigenesis but not quite reach the threshold to create full DNA mutations. Exposome is the combined exposure to all factors that cause internal chemical environment change[220]. Promoters, however, can change gene expression that leads uncontrolled growth. Initiators are factors that begin the process of DNA mutation and alteration in structure. It is not the exposure to one carcinogenic chemical or product that immediately causes DNA mutation and tumorigenesis, but it is the initial exposure that primes the tumor cells to change the function of certain genes. Eventually, the exposome can promote tumors to form[220]. It is also not just the exposure to one carcinogen in low doses but the repeated exposure to multiple carcinogens. In the case of cutaneous malignancies, solar UV is seen as a carcinogen that can either work as a promoter, an initiator, or both a promoter and initiator depending on the amount of exposure and protective measures. Moreover, studies have found that children exposed to the high-level UV from Western Australian sun have been found to have varying risk of cutaneous malignancies. Patients that arrive to Western Australia prior to age 10 have a 50% increased risk of dysplastic nevi or melanoma compared to those who arrive after the age of 10 regardless of their time spent sunbathing. This factor is due to the solar UV in Western Australia being higher of many other countries from where patients immigrate. Solar UV acting as an initiator and/or promoter for cutaneous malignancies explain the importance of protecting the skin from solar UV at all costs regardless of Fitzpatrick skin type. There is a need for further research to find other carcinogens that can act as initiators and/or promoters that to help patients prevent from these cancer-causing factors[221].
Effects of mi-RNAs
Genetic testing and gene therapy might be the future for patients with cutaneous malignancies. As previously stated, Castle Biosciences has already created testing to further determine staging and implication for sentinel node testing for melanoma and metastatic risk for SCC. However, the genetic testing does not stop there. miRNA plays an important role in signaling pathways determining the activation and suppression of specific genes for transcription factors. As mentioned in the previous sections, miRNAs can cause hypomethylation of genes leading to increased transcription, translation, and uncontrolled proliferation. Many of these genes are upregulated and downregulated having different effects on the genes; these effects can also lead to the appearance and progression of these cutaneous cancers. Moreover, SCC, VC, and CTCL have all been found to be incited by miRNAs. There are hundreds to thousands of miRNAs that can transform the function of genetic pathways and transcription factors from a normal functioning pathway to a malignant pathway. With this information, miRNAs can be a subject of research to help further test patients and possibly place them in complete remission as well as prophylaxis to prevent the next cutaneous malignancy from appearing.
Further genetic testing
In addition to genetic testing and therapy, there are many more topics within genetics that have room for further research. Many of the genetic tests performed are through somatic genetic testing and germline genetic testing. The germline testing is done through either saliva or lymphocytes. However, studies have found that within the saliva of patients with systematic lupus erythematosus, their salivary DNA has adapted the DNA of their food products[222]. “You are what you eat”. This information can be helpful; however, with determining the somatic genetic susceptibility of cutaneous malignancies, there must be a way to determine the difference between the genetic makeup of the food consumed and that of the patient’s unmutated, unincorporated somatic cell DNA and RNA. This can be a problem with lymphocytes as well. T- and B-lymphocytes have specific genetic makeup that turns them into the subtype of mature T- and B-lymphocytes. Yet, when T- and B-lymphocytes are presented with a specific antigen or allergen, their genetic makeup can also transform to program the lymphocyte for either attack of the antigen or produce antibodies towards the antigen, respectively. Additionally, lymphocytes can change their genetic code depending on their antigen priming, thus cancer cells may promote a change in lymphocyte DNA to avoid recognizing the cells as foreign[223]. There also will need to be consideration for the use of inactive mature B- and T-lymphocytes that have not yet been presented with antigen compared to that of the lymphocytes that have been presented with an antigen. It is possible that the human body is a state of active dynamic mosaicism with genes turning on and off that have adapted genetic material to food products and other antigens within the body. This information can ensure further accuracy when determining the somatic genetics of patients with cutaneous malignancies and help with their treatment and prognosis.
Cellular electrical charges
Another important factor to consider with skin cancer genetics is that tumors tend to also have an electrical charge as do many cells within the body. This is also a way to evade immune system attack. Tumor cells are negatively charged. Lymphocytes that have been primed with tumor antigen are also negatively charge. In the world of chemistry, similar charges are opposed to each other while opposite charges attract each other. However, antibodies are positively charged. The positive charge of antibodies assists with T-helper lymphocyte eradication of tumor cells. However, it is known that natural killer cells and CTLs are important in immune killing of tumor cells, so there is more research that needs to be done in terms of the effect of charge between immune cells and tumor cells as well as on genetic signaling.
Communication between immune system and nervous system
The central and peripheral nervous systems are important for regulating the systems in the body. In fact, the nervous system directly affects the function of the immune system against fighting malignancies. Chronic exposure to stressful events stimulates the adrenal gland, a neuroendocrine organ, to release sympathetic nervous system stimulating hormones which disturbs the normal function and balance of the immune system. In addition, this stress can also increase lymphatic vascularization in current tumors that increases the number of chemokines release and promotes diapedesis of tumor cells. It is important to note and follow patients who have chronic stressors without stress relief exercises to maintain homeostasis. This neuroendocrine modulation also can influence the appearance of cutaneous tumors. As a result, decreasing stressful events in the lives of patients can help prevent cutaneous malignancies from occurring[224].
The body as a flux state
It is important to remember that tumors are not always a collection of cells that function the same way undergoing the same processes at the same time. Many of these tumors function as organs, like that of the heart or lungs with various cell types and cell functions. Likewise, there can be tumor cells that are evading apoptosis by downregulating tumor suppressors, other cancer cells that are promoting cell growth through the mutation of oncogenes while others are invading the stroma by damaging the underlying adhesive molecules and releasing proteinases. In addition, cancer cells also can communicate amongst each other with autocrine and paracrine signaling. This signaling can activate specific genetic pathways to even mutate a normal, non-cancerous cell to that of a cancerous cell and change the genetic pathway of those that are already malignant. All these processes can occur simultaneously especially in the faster growing, more aggressive types of skin cancer. This hypothesis can explain why there are patients that can go into complete remission from a treatment type and others are treatment resistant. The multiple genetic signaling pathways activated, crosstalk and switching from one genetic pathway to another can be the reason as to why there is treatment resistance. Tumor cells, like immune cells, may have an ability to release chemokines leading to autocrine and paracrine communication between the cells. This process can also form a communication system between the cells telling each of them their function and even invade benign cells to become malignant. Adjuvant therapy might be considered in the earlier stages of cutaneous malignancies depending on the number of genetic pathways that are discovered within these tumor cells. These discoveries give providers further information to continuously perform full body skin examinations and surveillance patient chronic inflammatory conditions for transformation into malignant conditions. Furthermore, the incorporation of artificial intelligence data analysis of trends between the various pathways can help improve management of cutaneous malignancies by clinicians. Once these conditions are elucidated, oncologists can develop prospective personalized dynamic genetic testing for identifying the critical set of active oncogenetic pathways that need to be disrupted at particular moments. This process would then transform the lives of individual patients suffering from multiple cutaneous malignancies around the world.
Declarations
Authors’ contributionsWrote, final edited and revised the manuscript: Maner BS, Dupuis L, Su A, Jueng JJ, Harding TP, Meisenheimer VII J, Siddiqui FS, Hardack MR, Aneja S, Solomon JA
Contributed to Figure 10: Su A
Contributed to Figures 1, 2, 3, 6, 7, 8, 12 and reference organization: Meisenheimer VII J
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestNone that may be perceived as influencing the representation or interpretation of review other than Dr. James A. Solomon was a clinical investigator of Vismodegib clinical trials and expanded access program- funds paid to his employer and has been a paid consultant by Genentech, Sun Pharmaceuticals, Mayne Pharmaceuticals.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2020.
REFERENCES
2. Feehan RP, Shantz LM. Molecular signaling cascades involved in nonmelanoma skin carcinogenesis. Biochem J 2016;473:2973-94.
3. Sample A, He YY. Mechanisms and prevention of UV-induced melanoma. Photodermatol Photoimmunol Photomed 2018;34:13-24.
4. de Gruijl FR. Photocarcinogenesis: UVA vs. UVB radiation. Skin Pharmacol Appl Skin Physiol 2002;15:316-20.
6. Patrick MH. Studies on thymine-derived UV photoproducts in DNA--I. Formation and biological role of pyrimidine adducts in DNA. Photochem Photobiol 1977;25:357-72.
7. Lee JH, Budanov AV, Karin M. Sestrins orchestrate cellular metabolism to attenuate aging. Cell Metabolism 2013;18:792-801.
8. Budanov AV, Karin M. p53 target genes Sestrin1 and Sestrin2 connect genotoxic stress and mTOR signaling. Cell 2008;134:451-60.
9. Holick MF. Biological effects of sunlight, ultraviolet radiation, visible light, infrared radiation and vitamin D for health. Anticancer Res 2016;36:1345-56.
10. Leavitt E, Lask G, Martin S. Sonic hedgehog pathway inhibition in the treatment of advanced basal cell carcinoma. Curr Treat Options Oncol 2019;20:84.
11. Bolognia JJ, Jorizzo JL, Rapini RP. Dermatology. Philadelphia: Elsevier Saunders; 2012.
12. Xie J, Murone M, Luoh SM, Ryan A, Gu Q, et al. Activating smoothened mutations in sporadic basal-cell carcinoma. Nature 1998;391:90-2.
13. Marzuka AG, Book SE. Basal cell carcinoma: pathogenesis, epidemiology, clinical features, diagnosis, histopathology, and management. Yale J Biol Med 2015;88:167-79.
14. Gutzmer R, Solomon JA. Hedgehog pathway inhibition for the treatment of basal cell carcinoma. Target Oncol 2019;14:253-67.
15. Pala R, Alomari N, Nauli SM. Primary cilium-dependent signaling mechanisms. Int J Mol Sci 2017;18:2272.
16. Hutchin ME, Kariapper MS, Grachtchouk M, Wang A, Wei L, et al. Sustained hedgehog signaling is required for basal cell carcinoma proliferation and survival: conditional skin tumorigenesis recapitulates the hair growth cycle. Genes Dev 2005;19:214-23.
17. Pontén F, Berg C, Ahmadian A, Ren ZP, Nistér M, et al. Molecular pathology in basal cell cancer with p53 as a genetic marker. Oncogene 1997;15:1059-67.
18. Sekulic A, Migden MR, Oro AE, Dirix L, Lewis KD, et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N Engl J Med 2012;366:2171-9.
19. Kong JH, Siebold C, Rohatgi R. Biochemical mechanisms of vertebrate hedgehog signaling. Development (Cambridge, England) 2019;146:dev166892.
20. Bakkenist CJ, Kastan MB. DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature 2003;421:499-506.
21. Jazayeri A, Falck J, Lukas C, Bartek J, Smith GC, et al. ATM- and cell cycle-dependent regulation of ATR in response to DNA double-strand breaks. Nat Cell Biol 2006;8:37-45.
22. Shen T, Huang S. The role of Cdc25A in the regulation of cell proliferation and apoptosis. Anticancer Agents Med Chem 2012;12:631-9.
23. Decraene D, Agostinis P, Pupe A, de Haes P, Garmyn M. Acute response of human skin to solar radiation: regulation and function of the p53 protein. J Photochem Photobiol B 2001;63:78-83.
24. Liu P, Gan W, Chin YR, Ogura K, Guo J, et al. PtdIns(3,4,5)P3-dependent activation of the mTORC2 kinase complex. Cancer Discov 2015;5:1194-209.
25. Zhang Y, Hunter T. Roles of Chk1 in cell biology and cancer therapy. Int J Cancer 2014;134:1013-23.
26. Xiong Y, Hannon GJ, Zhang H, Casso D, Kobayashi R, et al. p21 is a universal inhibitor of cyclin kinases. Nature 1993;366:701-4.
27. Rezvani HR, Mazurier F, Cario-André M, Pain C, Ged C, et al. Protective effects of catalase overexpression on UVB-induced apoptosis in normal human keratinocytes. J Biol Chem 2006;281:17999-8007.
28. Sander CS, Hamm F, Elsner P, Thiele JJ. Oxidative stress in malignant melanoma and non-melanoma skin cancer. Br J Dermatol 2003;148:913-22.
29. Zhang J, Bowden GT. Activation of p38 MAP kinase and JNK pathways by UVA irradiation. Photochem Photobiol Sci 2012;11:54-61.
30. Nadeau PJ, Charette SJ, Toledano MB, Landry J. Disulfide bond-mediated multimerization of Ask1 and its reduction by thioredoxin-1 regulate H(2)O(2)-induced c-Jun NH(2)-terminal kinase activation and apoptosis. Mol Biol Cell 2007;18:3903-13.
31. Gross S, Knebel A, Tenev T, Neininger A, Gaestel M, et al. Inactivation of protein-tyrosine phosphatases as mechanism of UV-induced signal transduction. J Biol Chem 1999;274:26378-86.
32. Reinhardt HC, Aslanian AS, Lees JA, Yaffe MB. p53-deficient cells rely on ATM- and ATR-mediated checkpoint signaling through the p38MAPK/MK2 pathway for survival after DNA damage. Cancer Cell 2007;11:175-89.
33. Hess J, Angel P, Schorpp-Kistner M. AP-1 subunits: quarrel and harmony among siblings. J Cell Sci 2004;117:5965-73.
34. Silvers AL, Finch JS, Bowden GT. Inhibition of UVA-induced c-Jun N-terminal kinase activity results in caspase-dependent apoptosis in human keratinocytes. Photochem Photobiol 2006;82:423-31.
35. Cooper SJ, MacGowan J, Ranger-Moore J, Young MR, Colburn NH, et al. Expression of dominant negative c-jun inhibits ultraviolet B-induced squamous cell carcinoma number and size in an SKH-1 hairless mouse model. Mol Cancer Res 2003;1:848-54.
36. Strozyk E, Kulms D. The role of AKT/mTOR pathway in stress response to UV-irradiation: implication in skin carcinogenesis by regulation of apoptosis, autophagy and senescence. Int J Mol Sci 2013;14:15260-85.
37. Coffer PJ, Burgering BM, Peppelenbosch MP, Bos JL, Kruijer W. UV activation of receptor tyrosine kinase activity. Oncogene 1995;11:561-9.
38. Arora A, Scholar EM. Role of tyrosine kinase inhibitors in cancer therapy. J Pharmacol Exp Ther 2005;315:971-9.
39. Spallone G, Botti E, Costanzo A. Targeted therapy in nonmelanoma skin cancers. Cancers (Basel) 2011;3:2255-73.
41. Hafner C, Landthaler M, Vogt T. Activation of the PI3K/AKT signalling pathway in non-melanoma skin cancer is not mediated by oncogenic PIK3CA and AKT1 hotspot mutations. Exp Dermatol 2010;19:e222-7.
42. Ming M, He YY. PTEN: new insights into its regulation and function in skin cancer. J Invest Dermatol 2009;129:2109-12.
44. Shimobayashi M, Hall MN. Making new contacts: the mTOR network in metabolism and signalling crosstalk. Nat Rev Mol Cell Biol 2014;15:155-62.
45. Gan X, Wang J, Su B, Wu D. Evidence for direct activation of mTORC2 kinase activity by phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem 2011;286:10998-1002.
46. Tu Y, Ji C, Yang B, Yang Z, Gu H, et al. DNA-dependent protein kinase catalytic subunit (DNA-PKcs)-SIN1 association mediates ultraviolet B (UVB)-induced Akt Ser-473 phosphorylation and skin cell survival. Mol Cancer 2013;12:172.
47. Greer EL, Brunet A. FOXO transcription factors at the interface between longevity and tumor suppression. Oncogene 2005;24:7410-25.
48. Daitoku H, Fukamizu A. FOXO transcription factors in the regulatory networks of longevity. J Biochem 2007;141:769-74.
49. Bivik C, Ollinger K. JNK mediates UVB-induced apoptosis upstream lysosomal membrane permeabilization and Bcl-2 family proteins. Apoptosis 2008;13:1111-20.
50. Qin JZ, Bacon P, Panella J, Sitailo LA, Denning MF, et al. Low-dose UV-radiation sensitizes keratinocytes to TRAIL-induced apoptosis. J Cell Physiol 2004;200:155-66.
51. Kim DJ, Kataoka K, Sano S, Connolly K, Kiguchi K, et al. Targeted disruption of Bcl-xL in mouse keratinocytes inhibits both UVB- and chemically induced skin carcinogenesis. Mol Carcinog 2009;48:873-85.
52. Obsil T, Ghirlando R, Anderson DE, Hickman AB, Dyda F. Two 14-3-3 binding motifs are required for stable association of Forkhead transcription factor FOXO4 with 14-3-3 proteins and inhibition of DNA binding. Biochemistry 2003;42:15264-72.
54. Wellbrock C, Karasarides M, Marais R. The RAF proteins take centre stage. Nat Rev Mol Cell Biol 2004;5:875-85.
55. McCubrey JA, Steelman LS, Chappell WH, Abrams SL, Wong EW, et al. Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochim Biophys Acta 2007;1773:1263-84.
56. Omran OM, Ata HS. Expression of tumor necrosis factor-related apoptosis-inducing ligand death receptors DR4 and DR5 in human nonmelanoma skin cancer. Am J Dermatopathol 2014;36:710-7.
57. Shimizu T, Tolcher AW, Papadopoulos KP, Beeram M, Rasco DW, et al. The clinical effect of the dual-targeting strategy involving PI3K/AKT/mTOR and RAS/MEK/ERK pathways in patients with advanced cancer. Clin Cancer Res 2012;18:2316-25.
58. Paul G, Marchelletta RR, McCole DF, Barrett KE. Interferon-γ alters downstream signaling originating from epidermal growth factor receptor in intestinal epithelial cells: functional consequences for ion transport. J Biol Chem 2012;287:2144-55.
59. Chaisuparat R, Limpiwatana S, Kongpanitkul S, Yodsanga S, Jham BC. The Akt/mTOR pathway is activated in verrucous carcinoma of the oral cavity. J Oral Pathol Med 2016;45:581-5.
60. Kim C, Pasparakis M. Epidermal p65/NF-κB signalling is essential for skin carcinogenesis. EMBO Mol Med 2014;6:970-83.
61. Hoesel B, Schmid JA. The complexity of NF-κB signaling in inflammation and cancer. Mol Cancer 2013;12:86.
62. Carpenter RL, Lo HW. STAT3 target genes relevant to human cancers. Cancers (Basel) 2014;6:897-925.
63. Wilson NS, Dixit V, Ashkenazi A. Death receptor signal transducers: nodes of coordination in immune signaling networks. Nat Immunol 2009;10:348-55.
64. Gordon R. Skin cancer: an overview of epidemiology and risk factors. Semin Oncol Nurs 2013;29:160-9.
65. Burton KA, Ashack KA, Khachemoune A. Cutaneous squamous cell carcinoma: a review of high-risk and metastatic disease. Am J Clin Dermatol 2016;17:491-508.
66. Immunotherapy drug cemiplimab approved for advanced squamous cell skin cancer. Available from: https://www.cancer.gov/news-events/cancer-currents-blog/2018/cemiplimab-fda-squamous-cell-carcinoma. [Last accessed on 7 Sep 2020].
67. Benjamin CL, Ananthaswamy HN. p53 and the pathogenesis of skin cancer. Toxicol Appl Pharmacol 2007;224:241-8.
68. Kang D, Choi TH, Han K, Son D, Kim JH, et al. Regulation of K(+) channels may enhance wound healing in the skin. Med Hypotheses 2008;71:927-9.
69. Liu Q, Yu S, Zhao W, Qin S, Chu Q, et al. EGFR-TKIs resistance via EGFR-independent signaling pathways. Mol Cancer 2018;17:53.
70. Wenczak BA, Lynch JB, Nanney LB. Epidermal growth factor receptor distribution in burn wounds. Implications for growth factor-mediated repair. J Clin Invest 1992;90:2392-401.
71. Heo JS, Lee MY, Han HJ. Sonic hedgehog stimulates mouse embryonic stem cell proliferation by cooperation of Ca2+/protein kinase C and epidermal growth factor receptor as well as Gli1 activation. Stem Cells 2007;25:3069-80.
72. Carballo GB, Honorato JR, de Lopes GPF, Spohr TCLdSE. A highlight on Sonic hedgehog pathway. Cell Communication and Signaling 2018;16:11.
73. Panelos J, Tarantini F, Paglierani M, Di Serio C, Maio V, et al. Photoexposition discriminates Notch 1 expression in human cutaneous squamous cell carcinoma. Mod Pathol 2008;21:316-25.
74. Yang L, XL, Brooks YS. Dysregulated estrogen signaling through CYP1B1 contributes to notch deficiency in squamous cell carcinoma. Society of investigative dermatology: youtube presentation; 2020. Available from: https://www.youtube.com/watch?v=nxpbcf1xdca. [Last accessed on 7 Sep 2020].
75. Al Labban D, Jo SH, Ostano P, Saglietti C, Bongiovanni M, et al. Notch-effector CSL promotes squamous cell carcinoma by repressing histone demethylase KDM6B. J Clin Invest 2018;128:2581-99.
76. Konicke K, López-Luna A, Muñoz-Carrillo JL, Servín-González LS, Flores-de la Torre A, et al. The microRNA landscape of cutaneous squamous cell carcinoma. Drug Discov Today 2018;23:864-70.
77. Kollias N, Ruvolo E, Sayre RM. The value of the ratio of UVA to UVB in sunlight. Photochem Photobiol 2011;87:1474-5.
78. Mahler V. Skin diseases associated with environmental factors. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2017;60:605-17.
79. Agar N, Young AR. Melanogenesis: a photoprotective response to DNA damage? Mutat Res 2005;571:121-32.
80. Hung KF, Sidorova JM, Nghiem P, Kawasumi M. The 6-4 photoproduct is the trigger of UV-induced replication blockage and ATR activation. Proc Natl Acad Sci U S A 2020;117:12806-16.
81. Center V-IC. Cell cycle control: overview. Cell Cycle Control. Vanderbilt University; 2016.
82. Ra SH, Su A, Li X, Zhou J, Cochran AJ, et al. Keratoacanthoma and squamous cell carcinoma are distinct from a molecular perspective. Mod Pathol 2015;28:799-806.
83. Atasoy M, Anadolu-Braise R, Pirim I, Dogan H, Ikbal M. HLA antigen profile differences in patients with SCC (Squamous Cell Carcinoma) in-situ/actinic keratosis and invasive SCC: is there a genetic succeptibility for invasive SCC development? Eurasian J Med 2009;41:162-4.
84. Berhane T, Halliday GM, Cooke B, Barnetson RS. Inflammation is associated with progression of actinic keratoses to squamous cell carcinomas in humans. Br J Dermatol 2002;146:810-5.
85. Maru GB, Gandhi K, Ramchandani A, Kumar G. The role of inflammation in skin cancer. Adv Exp Med Biol 2014;816:437-69.
86. Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer 2009;9:798-809.
87. Scola N, Skrygan M, Wieland U, Kreuter A, Gambichler T. Altered gene expression in squamous cell carcinoma arising from congenital unilateral linear porokeratosis. Clin Exp Dermatol 2012;37:781-5.
88. Pi J, Diwan BA, Sun Y, Liu J, Qu W, et al. Arsenic-induced malignant transformation of human keratinocytes: involvement of Nrf2. Free Radic Biol Med 2008;45:651-8.
89. Colman JA. Arsenic and uranium in water from private wells completed in bedrock of east-central Massachusetts-Concentrations, correlations with bedrock units, and estimated probability maps. Sci Invest Rep 2011. doi: 10.3133/sir20115013
90. Li C, Srivastava RK, Elmets CA, Afaq F, Athar M. Arsenic-induced cutaneous hyperplastic lesions are associated with the dysregulation of Yap, a Hippo signaling-related protein. Biochem Biophys Res Commun 2013;438:607-12.
91. Poulalhon N, Dalle S, Balme B, Thomas L. Fast-growing cutaneous squamous cell carcinoma in a patient treated with vismodegib. Dermatology 2015;230:101-4.
92. Nissinen L, Farshchian M, Riihilä P, Kähäri VM. New perspectives on role of tumor microenvironment in progression of cutaneous squamous cell carcinoma. Cell Tissue Res 2016;365:691-702.
93. Zgraggen S, Huggenberger R, Kerl K, Detmar M. An important role of the SDF-1/CXCR4 axis in chronic skin inflammation. PLoS One 2014;9:e93665.
94. Sinha S, Su S, Workentine M, Agabalyan N, Cheng M, et al. Transcriptional analysis reveals evidence of chronically impeded ECM turnover and epithelium-to-mesenchyme transition in scar tissue giving rise to marjolin’s ulcer. J Burn Care Res 2017;38:e14-22.
95. Peters FS, Peeters AMA, Mandaviya PR, van Meurs JBJ, Hofland LJ, et al. Differentially methylated regions in T cells identify kidney transplant patients at risk for de novo skin cancer. Clin Epigenetics 2018;10:81.
96. Wysong A, Newman JG, Covington KR, Kurley SJ, Ibrahim SF, et al. Validation of a 40-gene expression profile test to predict metastatic risk in localized high-risk cutaneous squamous cell carcinoma. J Am Acad Dermatol 2020. doi: 10.1016/j.jaad.2020.04.088
97. Costache M, Desa LT, Mitrache LE, Pătraşcu OM, Dumitru A, et al. Cutaneous verrucous carcinoma - report of three cases with review of literature. Rom J Morphol Embryol 2014;55:383-8.
98. Schell BJ, Rosen T, Rády P, Arany I, Tschen JA, et al. Verrucous carcinoma of the foot associated with human papillomavirus type 16. J Am Acad Dermatol 2001;45:49-55.
99. Murao K, Kubo Y, Fukumoto D, Matsumoto K, Arase S. Verrucous carcinoma of the scalp associated with human papillomavirus type 33. Dermatol Surg 2005;31:1363-5.
100. Fujita S, Senba M, Kumatori A, Hayashi T, Ikeda T, et al. Human papillomavirus infection in oral verrucous carcinoma: genotyping analysis and inverse correlation with p53 expression. Pathobiology 2008;75:257-64.
101. Ren B. E2F integrates cell cycle progression with DNA repair, replication, and G2/M checkpoints. Genes Dev 2002;16:245-56.
102. Yim EK, Park JS. The role of HPV E6 and E7 oncoproteins in HPV-associated cervical carcinogenesis. Cancer Res Treat 2005;37:319.
103. Pătraşcu V, Geoloaica L, Ciurea R. Case report acral verrucous carcinoma. Curr Health Sci J 2019;45:235-40.
104. Schumann H, Roth W, Has C, Volz A, Erfurt-Berge C, et al. Verrucous carcinoma in epidermolysis bullosa simplex is possibly associated with a novel mutation in the keratin 5 gene. Br J Dermatol 2012;167:929-36.
105. Jung H, Seong HA, Ha H. Critical role of cysteine residue 81 of macrophage migration inhibitory factor (MIF) in MIF-induced inhibition of p53 activity. J Biol Chem 2008;283:20383-96.
106. Coulombe PA, Lee CH. Defining keratin protein function in skin epithelia: epidermolysis bullosa simplex and its aftermath. J Invest Dermatol 2012;132:763-75.
107. Bolling MC, Lemmink HH, Jansen GHL, Jonkman MF. Mutations in KRT5 and KRT14 cause epidermolysis bullosa simplex in 75% of the patients. Br J Dermatol 2011;164:637-44.
109. Deng Z, Wang Y, Fang X, Yan F, Pan H, et al. Research on miRNA-195 and target gene CDK6 in oral verrucous carcinoma. Cancer Gene Ther 2017;24:282-8.
110. Deng Z, Wang Y, Fang X, Yan F, Pan H, et al. Research on miRNA-195 and target gene CDK6 in oral verrucous carcinoma. Cancer Gene Ther 2017;24.
111. Adegboyega PA, Boromound N, Freeman DH. Diagnostic utility of cell cycle and apoptosis regulatory proteins in verrucous squamous carcinoma. Appl Immunohistochem Mol Morphol 2005;13:171-7.
112. Kusume T, Tsuda H, Kawabata M, Inoue T, Umesaki N, et al. The p16-Cyclin D1/CDK4-pRb pathway and clinical outcome in epithelial ovarian cancer. Clin Cancer Res 1999;5.
113. Al-Mohanna MA, Manogaran PS, Al-Mukhalafi Z, A Al-Hussein K, Aboussekhra A. The tumor suppressor p16INK4a gene is a regulator of apoptosis induced by ultraviolet light and cisplatin. Oncogene 2004;23:201-12.
114. Bruno S, Darzynkiewicz Z. Cell cycle dependent expression and stability of the nuclear protein detected by Ki-67 antibody in HL-60 cells. Cell Proliferation 1992;25:31-40.
115. Mallick S, Breta M, Gupta SD, Dinda AK, Mohanty BK, et al. Angiogenesis, proliferative activity and DNA ploidy in oral verrucous carcinoma: a comparative study including verrucous hyperplasia and squamous cell carcinoma. Pathol Oncol Res 2015;21:1249-57.
116. Yang G, Rosen DG, Liu G, Yang F, Guo X, et al. CXCR2 promotes ovarian cancer growth through dysregulated cell cycle, diminished apoptosis, and enhanced angiogenesis. Clin Cancer Res 2010;16:3875-86.
118. Langowski JL, Zhang X, Wu L, Mattson JD, Chen T, et al. IL-23 promotes tumour incidence and growth. Nature 2006;442:461-5.
119. Tchernev G, Guarneri C, Bevelacqua V, Wollina U. Carcinoma cuniculatum in course of etanercept: blocking autoimmunity but propagation of carcinogenesis? Int J Immunopathol Pharmacol 2014;27:261-6.
120. Chen Y, Sun J, Yang Y, Huang Y, Liu G. Malignancy risk of anti-tumor necrosis factor alpha blockers: an overview of systematic reviews and meta-analyses. Clin Rheumatol 2016;35:1-18.
122. Basosquamous Cell Cancer. In: Hoffman H, editor. Iowa Head and Neck Protocols: University of Iowa Health Care; 2017.
123. Akay BN, Saral S, Heper AO, Erdem C, Rosendahl C. Basosquamous carcinoma: Dermoscopic clues to diagnosis. J Dermatol 2017;44:127-34.
124. Anand RL, Collins D, Chapman A. Basosquamous carcinoma: appearance and reality. Oxf Med Case Rep 2017;2017.
125. Tarapore E, Atwood SX. Defining the genetics of basosquamous carcinoma. J Invest Dermatol 2019;139:2258-60.
126. Murthaiah P, Truskinovsky A, Shah S, Dudek A. Collision tumor versus multiphenotypic differentiation: a case of carcinoma with features of colonic and lung primary tumors. Anticancer Res 2009;29:1495-7.
127. Lara F, Santamaría JR, Garbers LM. Recurrence rate of basal cell carcinoma with positive histopathological margins and related risk factors. An Bras Dermatol 2017;92:58-62.
128. Oldbury J, Wain R, Abas S, Dobson C, Iyer S. Basosquamous carcinoma: a single centre clinicopathological evaluation and proposal of an evidence-based protocol. J Skin Cancer 2018;2018.
129. Piva de Freitas P, Senna CG, Tabai M, Chone CT, Altemani A. Metastatic basal cell carcinoma: a rare manifestation of a common disease. Case Rep Med 2017;2017.
130. Major A, Anderson M. Not just skin deep: distant metastases from cutaneous squamous cell carcinoma. Am J Med 2017;130.
131. Alam M. Basal cell carcinoma recurrence after mohs surgery. U.S. National Laboratory of Medicine; 2012.
132. Metastatic Squamous Cell Carcinoma. Transplant Skin Cancer Network: University of California San Francisco. Available from: https://skincancer.ucsf.edu/metastatic-squamous-cell-carcinoma. [Last accessed on 7 Sep 2020].
133. van Lee CB, Roorda BM, Wakkee M, Voorham Q, Mooyaart AL, et al. Recurrence rates of cutaneous squamous cell carcinoma of the head and neck after Mohs micrographic surgery vs. standard excision: a retrospective cohort study. Br J Dermatol 2019;181:338-43.
134. Martin RC 2nd, Edwards MJ, Cawte TG, Sewell CL, McMasters KM. Basosquamous carcinoma: analysis of prognostic factors influencing recurrence. Cancer 2000;88:1365-9.
135. Chiang A, Tan CZ, Kuonen F, Hodgkinson LM, Chiang F, et al. Genetic mutations underlying phenotypic plasticity in basosquamous carcinoma. J Invest Dermatol 2019;139:2263-71.e5.
136. Zhao X, Ponomaryov T, Ornell KJ, Zhou P, Dabral SK, et al. RAS/MAPK activation drives resistance to smo inhibition, metastasis, and tumor evolution in shh pathway-dependent tumors. Cancer Res 2015;75:3623-35.
137. Osawa H, Ohnishi H, Takano K, Noguti T, Mashima H, et al. Sonic hedgehog stimulates the proliferation of rat gastric mucosal cells through ERK activation by elevating intracellular calcium concentration. BiochemBiophys Res Commun 2006;344:680-7.
138. Solomon JA, Iarrobino A, Shutty B. Calcium dependence of the SHH pathway and theoretical implications in oral treatment of locally advanced or metastatic basal cell carcinomas. Cutaneous Oncol Today 2011:9-13.
139. Ji J, Wernli M, Mielgo A, Buechner SA, Erb P. Fas-ligand gene silencing in basal cell carcinoma tissue with small interfering RNA. Gene Therapy 2005;12:678-84.
140. Hardwick JM, Soane L. Multiple functions of BCL-2 family proteins. Cold Spring Harb Perspect Biol 2013;5.
141. N Sivrikoz O, Kandiloğlu G. The effects of cyclin D1 and Bcl-2 expressıon on aggressive behavior in basal cell and basosquamous carcinoma. Iran J Pathol 2015;10:185-91.
142. Rishikaysh P, Dev K, Diaz D, Qureshi WMS, Filip S, et al. Signaling involved in hair follicle morphogenesis and development. Int J Mol Sci 2014;15:1648-70.
143. Ellis T, Smyth I, Riley E, Bowles J, Adolphe C, et al. Overexpression of sonic hedgehog suppresses embryonic hair follicle morphogenesis. Dev Biol 2003;263:203-15.
144. Kasper M, Jaks V, Are A, Bergström Å, Schwäger A, et al. Wounding enhances epidermal tumorigenesis by recruiting hair follicle keratinocytes. Proc Nat Acad Sci 2011;108:4099-104.
145. Peterson SC, Eberl M, Vagnozzi AN, Belkadi A, Veniaminova NA, et al. Basal cell carcinoma preferentially arises from stem cells within hair follicle and mechanosensory niches. Cell Stem Cell 2015;16:400-12.
146. Gutzmer R, Solomon J. Hedgehog pathway inhibition for the treatment of basal cell carcinoma. Target Oncol 2019;14:253-67.
147. Duman N, Şen Korkmaz N, Erol Z. Host immune responses and peritumoral stromal reactions in different basal cell carcinoma subtypes: histopathological comparison of basosquamous carcinoma and high-risk and low-risk basal cell carcinoma subtypes. Turk J Med Sci 2016;46:28-34.
148. Zainab H, Sultana A, Shaimaa. Stromal desmoplasia as a possible prognostic indicator in different grades of oral squamous cell carcinoma. J Oral Maxillofac Pathol 2019;23:338-43.
149. Kaporis H, Guttman-Yassky E, Lowes M, Haider A, Fuentes-Dunclan J, et al. Human basal cell carcinoma is associated with Foxp3+ T cells in a Th2 dominant microenvironment. J Invest Dermatol 2007;127:2391-8.
151. Skin Cancer Facts & Statistics. 2020. Available from: https://www.skincancer.org/skin-cancer-information/skin-cancer-facts/. [Last accessed on 7 Sep 2020].
152. Wells GL. Melanoma - Dermatologic Disorders. Merck Manuals Professional Edition 2019. Available from: https://www.msdmanuals.com/professional/dermatologic-disorders/cancers-of-the-skin/melanoma. [Last accessed on 7 Sep 2020].
153. Soufir N. Prevalence of p16 and CDK4 germline mutations in 48 melanoma-prone families in France. The French Familial Melanoma Study Group. Hum Mol Genet 1998;7:209-16.
154. Freedman DM, Sigurdson A, Rao RS, Hauptmann M, Alexander B, et al. Risk of melanoma among radiologic technologists in the United States. Int J Cancer 2003;103:556-62.
155. Miura K, Olsen CM, Rea S, Marsden J, Green AC. Do airline pilots and cabin crew have raised risks of melanoma and other skin cancers? Systematic review and meta-analysis. Br J Dermatol 2019;181:55-64.
156. Kubica AW, Brewer JD. Melanoma in immunosuppressed patients. Mayo Clinic Proceedings 2012;87:991-1003.
157. Budden T, Davey RJ, Vilain RE, Ashton KA, Braye SG, et al. Repair of UVB-induced DNA damage is reduced in melanoma due to low XPC and global genome repair. Oncotarget 2016;7:60940-53.
158. Serrano M, Lee HW, Chin L, Cordon-Cardo C, Beach D, et al. Role of the INK4a locus in tumor suppression and cell mortality. Cell 1996;85:27-37.
160. Palmieri G, Ombra M, Colombino M, Casula M, Sini M, et al. Multiple molecular pathways in melanomagenesis: characterization of therapeutic targets. Front Oncol 2015;5:183.
161. Liu J, Fukunaga-Kalabis M, Li L, Herlyn M. Developmental pathways activated in melanocytes and melanoma. Arch Biochem Biophys 2014;563:13-21.
162. Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell 2015;161:1681-96.
163. Curtin JA, Fridlyand J, Kageshita T, Patel HN, Busam KJ, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med 2005;353:2135-47.
164. Lopez-Bergami P, Fitchman B, Ronai ZE. Understanding signaling cascades in melanoma. Photochem Photobiol 2008;84:289-306.
165. Kunz M, Vera J. Modelling of protein kinase signaling pathways in melanoma and other cancers. Cancers 2019;11:465.
166. Paluncic J, Kovacevic Z, Jansson PJ, Kalinowski D, Merlot AM, et al. Roads to melanoma: key pathways and emerging players in melanoma progression and oncogenic signaling. Biochim Biophys Acta 2016;1863:770-84.
167. Orouji E, Orouji A, Gaiser T, Larribère L, Gebhardt C, et al. MAP kinase pathway gene copy alterations inNRAS/BRAFwild-type advanced melanoma. Int J Cancer 2016;138:2257-62.
168. Hodis E, Watson IR, Kryukov GV, Arold ST, Imielinski M, et al. A landscape of driver mutations in melanoma. Cell 2012;150:251-63.
169. Chen X, Wu Q, Depeille P, Chen P, Thornton S, et al. RasGRP3 mediates MAPK pathway activation in GNAQ mutant uveal melanoma. Cancer Cell 2017;31:685-96.e6.
170. Davies H, Bignell GR, Cox C, Stephens P, Edkins S, et al. Mutations of the BRAF gene in human cancer. Nature 2002;417:949-54.
171. Broussard L, Howland A, Ryu S, Song K, Norris D, et al. Melanoma cell death mechanisms. Chonnam Med J 2018;54:135.
172. Abbaspour Babaei M, Kamalidehghan B, Saleem M, Zaman Huri H, Ahmadipour F. Receptor tyrosine kinase (c-Kit) inhibitors: a potential therapeutic target in cancer cells. Drug Des Devel Ther 2016;10:2443-59.
173. Meng D, Carvajal RD. KIT as an oncogenic driver in melanoma: an update on clinical development. Am J Clin Dermatol 2019;20:315-23.
174. Sakaizawa K, Ashida A, Uchiyama A, Ito T, Fujisawa Y, et al. Clinical characteristics associated with BRAF, NRAS and KIT mutations in Japanese melanoma patients. J Dermatol Sci 2015;80:33-7.
175. Kong Y, Si L, Zhu Y, Xu X, Corless CL, et al. Large-scale analysis of KIT aberrations in Chinese patients with melanoma. Clin Cancer Res 2011;17:1684-91.
176. Avagliano A, Fiume G, Pelagalli A, Sanità G, Ruocco MR, et al. Metabolic plasticity of melanoma cells and their crosstalk with tumor microenvironment. Front Oncol 2020;10:722.
177. Hartman ML, Czyz M. MITF in melanoma: mechanisms behind its expression and activity. Cell Mol Life Sci 2015;72:1249-60.
178. Wellbrock C, Arozarena I. The complexity of the ERK/MAP-kinase pathway and the treatment of melanoma skin cancer. Front cell dev biol 2016;4:33.
179. Buscà R, Berra E, Gaggioli C, Khaled M, Bille K, et al. Hypoxia-inducible factor 1{alpha} is a new target of microphthalmia-associated transcription factor (MITF) in melanoma cells. J Cell Biol 2005;170:49-59.
180. McGill GG, Horstmann M, Widlund HR, Du J, Motyckova G, et al. Bcl2 regulation by the melanocyte master regulator mitf modulates lineage survival and melanoma cell viability. Cell 2002;109:707-18.
181. Šestáková B, Ondrušová L, Vachtenheim J. Cell cycle inhibitor p21/WAF1/CIP1 as a cofactor of MITF expression in melanoma cells. Pigment Cell Melanoma Res 2010;23:238-51.
182. McKibbin T. Melanoma: understanding relevant molecular pathways as well as available and emerging therapies. Am J Manag Care 2015;21:S224-33.
183. Wei SC, Levine JH, Cogdill AP, Zhao Y, Anang NAAS, et al. Distinct cellular mechanisms underlie anti-CTLA-4 and anti-PD-1 checkpoint blockade. Cell 2017;170:1120-33.e17.
184. Sznol M, Chen L. Antagonist antibodies to PD-1 and B7-H1 (PD-L1) in the treatment of advanced human cancer. Clin Cancer Res 2013;19:1021-34.
185. Zhang N, Wang L, Zhu G, Sun DJ, He H, et al. The association between trauma and melanoma in the Chinese population: a retrospective study. J Eur Acad Dermatol Venereol 2013;28.
187. Elmore JG, Elder DE, Barnhill RL, Knezevich SR, Longton GM, et al. Concordance and reproducibility of melanoma staging according to the 7th vs 8th edition of the AJCC cancer staging manual. JAMA Network Open 2018;1:e180083.
188. Gastman BR, Gerami P, Kurley SJ, Cook RW, Leachman S, et al. Identification of patients at risk of metastasis using a prognostic 31-gene expression profile in subpopulations of melanoma patients with favorable outcomes by standard criteria. J Am Acad Dermatol 2019;80:149-57.e4.
189. Gerami P, Cook RW, Wilkinson J, Russell MC, Dhillon N, et al. Development of a prognostic genetic signature to predict the metastatic risk associated with cutaneous melanoma. Clin Cancer Res 2015;21:175-83.
190. Gastman BR, Zager JS, Messina JL, Cook RW, Covington KR, et al. Performance of a 31-gene expression profile test in cutaneous melanomas of the head and neck. Head Neck 2019;41:871-9.
191. Greenhaw BN, Covington KR, Kurley SJ, Yeniay Y, Cao NA, et al. Molecular risk prediction in cutaneous melanoma: a meta-analysis of the 31-gene expression profile prognostic test in 1,479 patients. J Am Acad Dermatol 2020. doi: 10.1016/j.jaad.2020.03.053
192. Kovarik CL, Chu EY, Adamson AS. Gene expression profile testing for thin melanoma: evidence to support clinical use remains thin. JAMA Dermatol 2020. doi: 10.1001/jamadermatol.2020.0894
193. Dalal M, Mitchell S, McCloskey C, Zagadailov E, Gautam A. The clinical and humanistic burden of cutaneous T-cell lymphomas and response to conventional and novel therapies: results of a systematic review. Expert Rev Hematol 2020;13:405-19.
195. Jahan-Tigh RR, Huen AO, Lee GL, Pozadzides JV, Liu P, et al. Hydrochlorothiazide and cutaneous T cell lymphoma. Cancer 2013;119:825-31.
196. Emge DA, Bassett RL, Duvic M, Huen AO. Methicillin-resistant Staphylococcus aureus (MRSA) is an important pathogen in erythrodermic cutaneous T-cell lymphoma (CTCL) patients. Arch Dermatol Res 2020;312:283-8.
198. Rasheed H, Hegazy RA, Gawdat HI, Mehaney DA, Kamel MM, et al. Serum vitamin D and vitamin D receptor gene polymorphism in mycosis fungoides patients: a case control study. PLoS One 2016;11:e0158014.
199. Choi J, Goh G, Walradt T, Hong BS, Bunick CG, et al. Genomic landscape of cutaneous T cell lymphoma. Nat Genet 2015;47:1011-9.
200. Ayhan A, Mao TL, Suryo Rahmanto Y, Zeppernick F, Ogawa H, et al. Increased proliferation in atypical hyperplasia/endometrioid intraepithelial neoplasia of the endometrium with concurrent inactivation of ARID1A and PTEN tumour suppressors. J Pathol Clin Res 2015;1:186-93.
201. Yang L, Rau R, Goodell MA. DNMT3A in haematological malignancies. Nature Reviews Cancer 2015;15:152-65.
202. Gallardo F, Sandoval J, Díaz-Lagares A, Garcia R, D’Altri T, et al. Notch1 pathway activation results from the epigenetic abrogation of notch-related micrornas in mycosis fungoides. J Invest Dermatol 2015;135:3144-52.
203. Dotto GP. Crosstalk of Notch with p53 and p63 in cancer growth control. Nat Rev Cancer 2009;9:587-95.
204. Espinosa L, Cathelin S, D’Altri T, Trimarchi T, Statnikov A, et al. The Notch/Hes1 pathway sustains NF-κB Activation through CYLD repression in T cell leukemia. Cancer Cell 2010;18:268-81.
205. Manfè V, Biskup E, Rosbjerg A, Kamstrup M, Skov AG, et al. miR-122 regulates p53/Akt signalling and the chemotherapy-induced apoptosis in cutaneous T-cell lymphoma. PLoS One 2012;7:e29541.
206. Walia R, Yeung CCS. An update on molecular biology of cutaneous T cell lymphoma. Front Oncol 2020;9:1558.
207. Wahnschaffe L, Braun T, Timonen S, Giri AK, Schrader A, et al. JAK/STAT-activating genomic alterations are a hallmark of T-PLL. Cancers 2019;11:1833.
208. Rawlings JS, Rosler KM, Harrison DA. The JAK/STAT signaling pathway. J Cell Sci 2004;117:1281-3.
209. Ungewickell A, Bhaduri A, Rios E, Reuter J, Lee CS, et al. Genomic analysis of mycosis fungoides and Sézary syndrome identifies recurrent alterations in TNFR2. Nat Genet 2015;47:1056-60.
210. Espinosa L, Cathelin S, D’Altri T, Trimarchi T, Statnikov A, et al. The Notch/Hes1 pathway sustains NF-κB activation through CYLD repression in T cell leukemia. Cancer Cell 2010;18:268-81.
211. Vaqué JP, Gómez-López G, Monsálvez V, Varela I, Martínez N, et al. PLCG1 mutations in cutaneous T-cell lymphomas. Blood 2014;123:2034-43.
212. Pomerantz JL, Denny EM, Baltimore D. CARD11 mediates factor-specific activation of NF-κB by the T cell receptor complex. EMBO J 2002;21:5184-94.
213. Braun FCM, Grabarczyk P, Möbs M, Braun FK, Eberle J, et al. Tumor suppressor TNFAIP3 (A20) is frequently deleted in Sézary syndrome. Leukemia 2011;25:1494-501.
214. McGirt LY, Degesys CA, Johnson VE, Zic JA, Zwerner JP, et al. TOX expression and role in CTCL. J Eur Acad Dermatol Venereol 2016;30:1497-502.
215. Kremer KN, Dinkel BA, Sterner RM, Osborne DG, Jevremovic D, et al. TCR-CXCR4 signaling stabilizes cytokine mRNA transcripts via a PREX1-Rac1 pathway: implications for CTCL. Blood 2017;130:982-94.
216. Tanase C, Popescu I, Enciu AM, Gheorghisan-Galateanu A, Codrici E, et al. Angiogenesis in cutaneous T-cell lymphoma - proteomic approaches (Review). Oncol Lett 2019;17:4060-7.
217. Liszewski W, Naym DG, Biskup E, Gniadecki R. Psoralen with ultraviolet A-induced apoptosis of cutaneous lymphoma cell lines is augmented by type I interferons via the JAK1-STAT1 pathway. Photodermatol Photoimmunol Photomed 2017;33:164-71.
218. Shea L, Mehta-Shah N. Brentuximab vedotin in the treatment of peripheral T cell lymphoma and cutaneous T cell lymphoma. Curr Hematol Malig Rep 2020;15:9-19.
219. Jawed SI, Myskowski PL, Horwitz S, Moskowitz A, Querfeld C. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): Part II. Prognosis, management, and future directions. J Am Acad Dermatol 2014;70:223.e1-17.
221. English DR, Armstrong BK. Identifying people at high risk of cutaneous malignant melanoma: results from a case-control study in Western Australia. Br Med J (Clin Res Ed) 1988;296:1285-8.
222. Rizzi A, Raddadi N, Sorlini C, Nordgrd L, Nielsen KM, et al. The stability and degradation of dietary DNA in the gastrointestinal tract of mammals: implications for horizontal gene transfer and the biosafety of GMOs. Crit Rev Food Sci Nutr 2012;52:142-61.
223. Potter VR. Initiation and promotion in cancer formation: the importance of studies on intercellular communication. Yale J Biol Med 1980;53:367-84.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Maner BS, Dupuis L, Su A, Jueng JJ, Harding TP, Meisenheimer J, Siddiqui FS, Hardack MR, Aneja S, Solomon JA. Overview of genetic signaling pathway interactions within cutaneous malignancies. J Cancer Metastasis Treat 2020;6:37. http://dx.doi.org/10.20517/2394-4722.2020.60
AMA Style
Maner BS, Dupuis L, Su A, Jueng JJ, Harding TP, Meisenheimer J, Siddiqui FS, Hardack MR, Aneja S, Solomon JA. Overview of genetic signaling pathway interactions within cutaneous malignancies. Journal of Cancer Metastasis and Treatment. 2020; 6: 37. http://dx.doi.org/10.20517/2394-4722.2020.60
Chicago/Turabian Style
Maner, Brittany S., Leonie Dupuis, Ashley Su, Jeremy J Jueng, Tanner P. Harding, John Meisenheimer, Fahad S. Siddiqui, Mia R. Hardack, Savina Aneja, James A. Solomon. 2020. "Overview of genetic signaling pathway interactions within cutaneous malignancies" Journal of Cancer Metastasis and Treatment. 6: 37. http://dx.doi.org/10.20517/2394-4722.2020.60
ACS Style
Maner, BS.; Dupuis L.; Su A.; Jueng JJ.; Harding TP.; Meisenheimer J.; Siddiqui FS.; Hardack MR.; Aneja S.; Solomon JA. Overview of genetic signaling pathway interactions within cutaneous malignancies. J. Cancer. Metastasis. Treat. 2020, 6, 37. http://dx.doi.org/10.20517/2394-4722.2020.60
About This Article
Copyright
Data & Comments
Data

 Cite This Article 57 clicks
Cite This Article 57 clicks


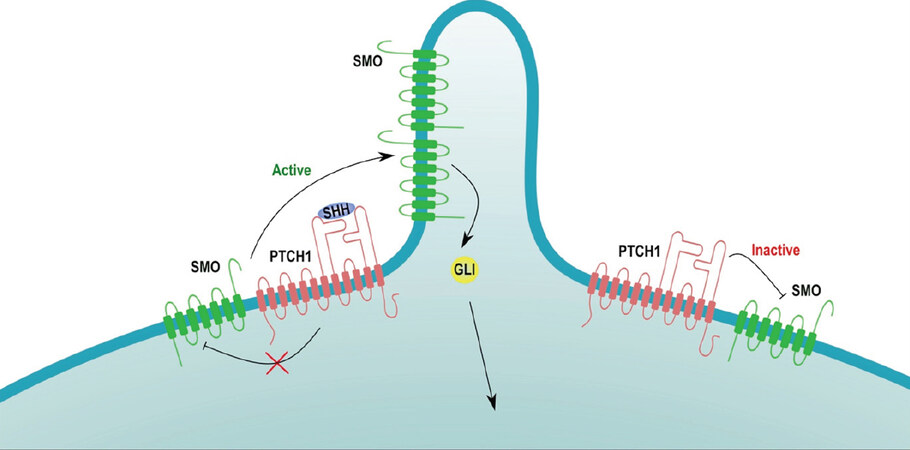
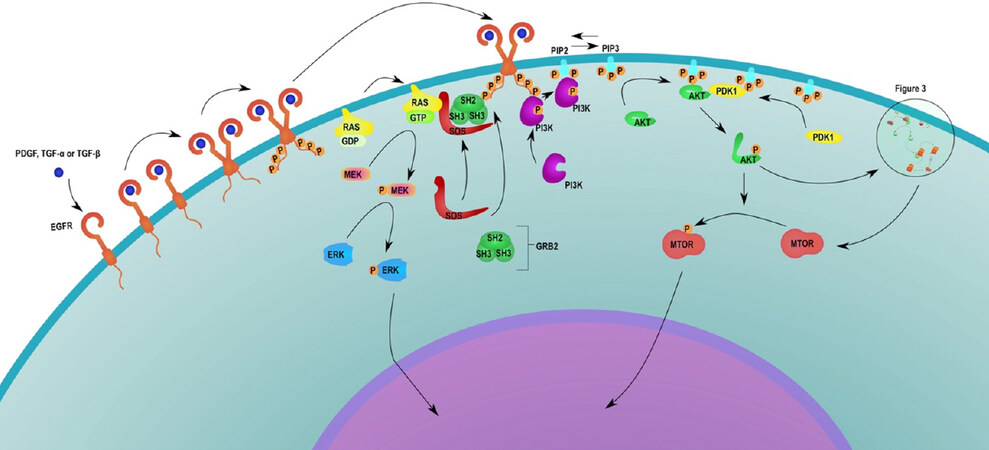
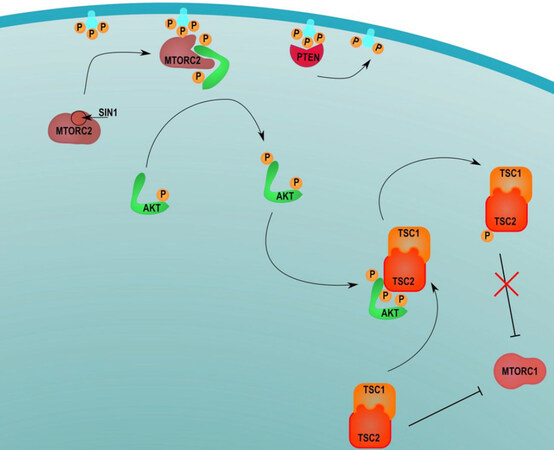
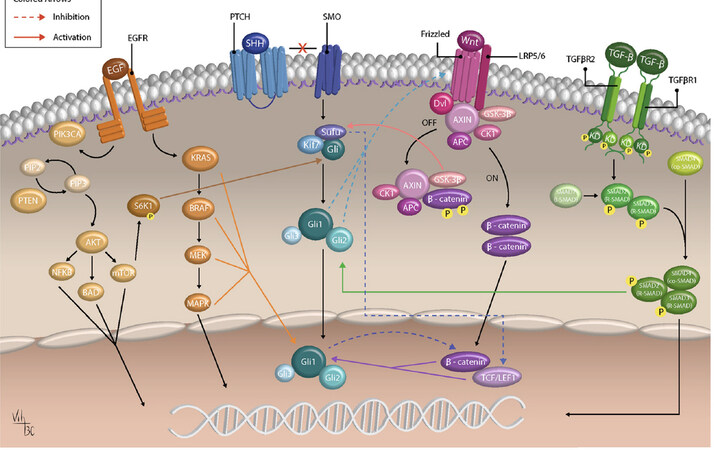
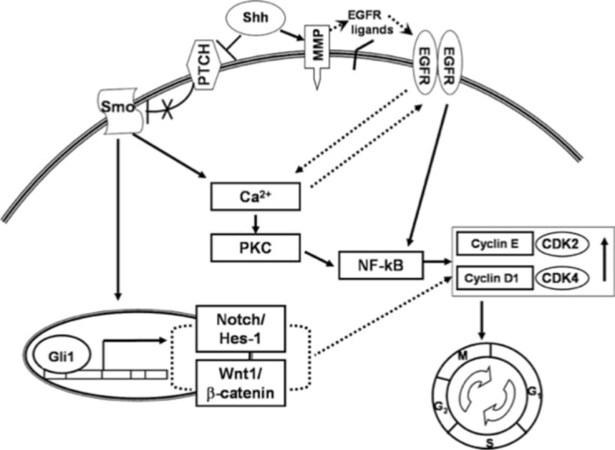
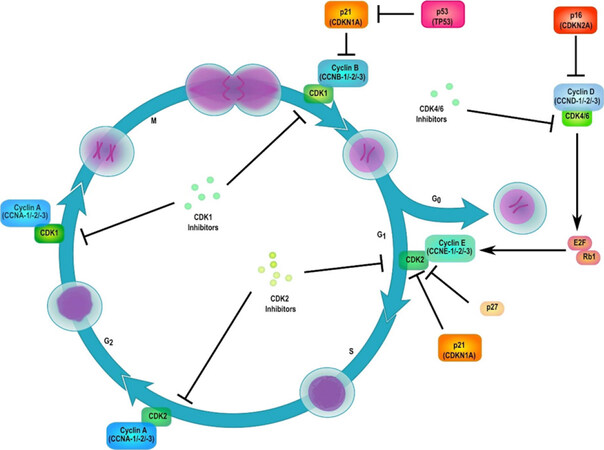
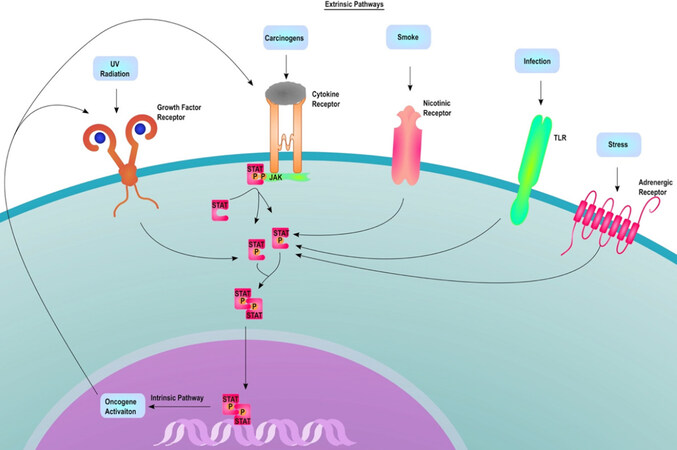
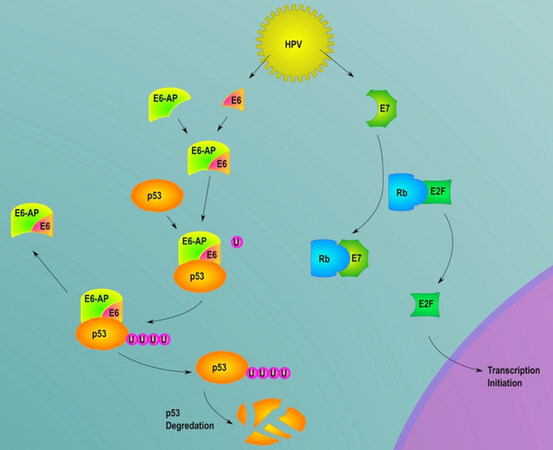
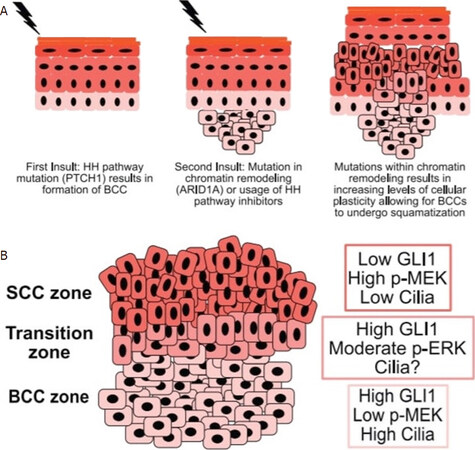
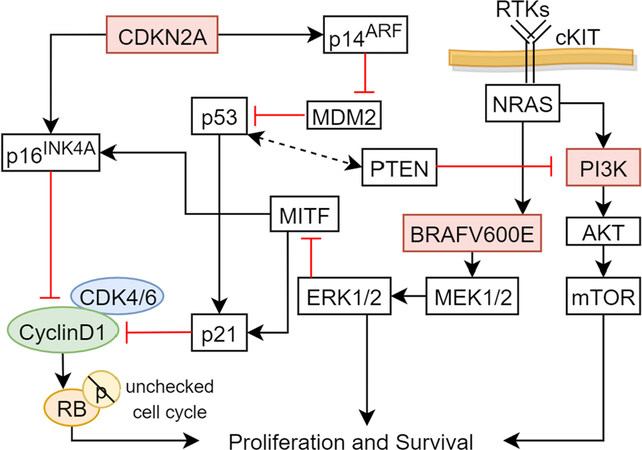
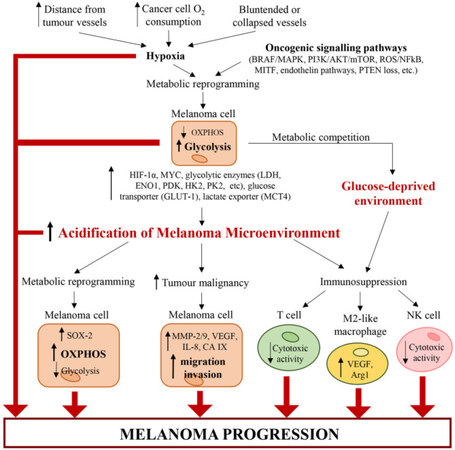









Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.