Gliomatosis cerebri: a monocentric real-life experience
Abstract
Aim: Gliomatosis cerebri (GC) is defined as a rare pattern of growth of diffuse gliomas involving three or more cerebral lobes. Given its rarity, it is difficult to define prognostic factors and standard of treatment. We retrospectively analyzed patients (PT) with GC from a single institution with the aim of identifying the main prognostic factors and to assess optimal management.
Methods: Medical records were reviewed of patients ≥ 18 years with a histological and/or radiological diagnosis of GC (with no contrast enhancement) occurring between 2006 and 2017. Median progression free survival (PFS) and overall survival (OS) were calculated by the Kaplan-Meier method.
Results: We analyzed 33 PT, 22 males and 11 females; Eastern Cooperative Oncology Group (ECOG) performance status (PS) was 0-1 in 21 of the patients. Twenty-two PT underwent biopsy: 16 were astrocytomas and 6 oligodendrogliomas. O6-methylguanin-DNA-methyltransferase (MGMT) was detected in 14 cases, and it was methylated in eight cases. Isocitrate dehydrogenase 1 (IDH1) was analyzed in 16 PT, and it had mutated in 10 of them. Nine PT (27%) were treated with radiation therapy (RT) plus concurrent temozolomide (TMZ), 22 PT (67%) received TMZ alone, and 2 PT (6%) underwent RT alone. We reported “complete response” in 1 patient (3%), partial response in 9 PT (27%), and stable disease in 15 PT (45%), while 8 PT (25%) had a progressive disease. For all PT, PFS and OS were 19.1 and 30.7 months, respectively. For ECOG PS 0-1 and ≥ 2, PFS was 34.6 months vs. 3.4 months (P < 0.0001) and OS was 42 months vs. 8.9 months (P < 0.0001), respectively. Methylated MGMT was associated with longer PFS (41.6 months vs. 8.9 months, P = 0.05) and OS (52.7 months vs. 14.6 months, P = 0.009); PFS for IDH1 mutation and IDH wild-type was 52.7 months vs. 8.9 months (P = 0.006) and OS was 52.7 months vs. 41.7 months (P = 0.02), respectively. No significant difference was detected as regards treatments. With regard to histological subtype, OS was 42.0 months vs. 52.7 months (P = 0.8) and PFS was 41.6 months vs. 28.6 months (P = 0.7) for astrocytoma vs. oligodendroglioma, respectively. PT with treatment response showed a longer OS. PT receiving second-line treatment had a longer OS of 30.7 months vs. 6.5 months (P = 0.04).
Conclusion: ECOG PS, MGMT methylation, and IDH1 mutational status seem to have an important prognostic significance, while the type of treatment does not seem to affect survival. Treatment response could be a surrogate marker for survival.
Keywords
INTRODUCTION
According to the 2016 World Health Organization (WHO) Classification of tumors of the Central Nervous System, gliomatosis cerebri is no longer a distinct pathological entity, being designated as a pattern of diffuse and extensive growth of glioma cells. It includes astrocytic and oligodendroglial tumors, involving three or more cerebral lobes, usually bilateral, with a frequent extension to infratentorial structures[1].
However, GC remains a complicated entity to treat, with great heterogeneity in clinical features and outcome. Symptoms and radiological appearance are non-specific, and, thus, it can be confused with other neurological diseases[2]; on magnetic resonance imaging (MRI), GC usually shows diffuse infiltration predominantly of white matter, with T2-weighted and fluid-attenuated inversion recovery (FLAIR) hyperintensity.
With regard to the treatment of GC, optimal management is still unclear. Surgery is used for focal biopsy and the role of radiotherapy and chemotherapy is discordant.
The aim of our paper is to describe our real-life experience in GC by retrospective analysis and to investigate the main prognostic factors and therapeutic management.
METHODS
We retrospectively analyzed all consecutive patients diagnosed with GC who visited our oncological center, the Veneto Institute of Oncology-IRCCS, in Padua between January 2006 and December 2017. Each patient had to meet the following criteria: age ≥ 18 years, MRI-T2 or FLAIR sequences showing the interest of at least three cerebral lobes, and with the aspect of diffuse glioma with no contrast enhancement. Histological diagnoses were based on the 2009 or 2016 WHO classification, depending on the year in which the intervention was performed.
Demographic data, age at diagnosis, biopsy execution, histological diagnosis, O6-methylguanin-DNA-methyltransferase (MGMT) promoter methylation, IDH1-2 mutation, type of therapy, radiological aspects, and response and patient outcomes were recorded. In particular, the MGMT promoter methylation status was investigated by polymerase chain reaction (PCR) or pyrosequencing and the IDH1-2 mutation status was performed by immunohistochemistry and PCR; radiological response was retrospectively evaluated by an expert neuroradiologist, according to the Response Assessment in Neuro-Oncology (RANO) criteria for diffuse low-grade gliomas[3]; patient outcomes were described in terms of progression free survival (PFS) and overall survival (OS).
As regards statistical analysis, PSF was defined as the time between diagnosis and clinical or radiological progression or death, and OS was defined as the time between diagnosis and patient death; both PFS and OS were calculated using Kaplan-Meier survival curves. Univariate analyses were performed for the Eastern Cooperative Oncology Group (ECOG) performance status (PS), MGMT promoter methylation, IDH1 mutation, type of first-line treatment [RT + temozolomide (TMZ), RT alone, or TMZ alone], histological subtype (astrocytoma vs. oligodendroglioma), best response to treatment [stable disease (SD), partial response (PR), complete response (CR), or progressive disease], and second-line treatment (yes vs. no).
RESULTS
After reviewing patient records, 33 patients were eligible for the study. Patient features and tumor characteristics are summarized in Table 1.
Patient, tumor, and treatment characteristics
| N (%) | |
| Patients | 33 |
| Sex | |
| Male | 22 (67) |
| Female | 11 (33) |
| ECOG PS | |
| 0-1 | 21 (64) |
| ≥ 2 | 12 (36) |
| Biopsy | |
| Yes | 22 (67) |
| No | 11 (33) |
| Histology | |
| Astrocytoma | 16 (73) |
| Oligodendroglioma | 6 (27) |
| MGMT status | |
| Methylated | 8/14 (57) |
| Unmethylated | 6/14 (43) |
| IDH status | |
| Mutated | 10/16 (63) |
| Wild-type | 6/16 (37) |
| Type of treatment | |
| RT + TMZ | 9 (27) |
| TMZ alone | 22 (67) |
| RT alone | 2 (6) |
| Radiological response | |
| CR | 1 (3) |
| PR | 9 (27) |
| SD | 15 (45) |
| PD | 8 (25) |
| Second-line treatment | 13 (59) |
| RT + TMZ | 1 (5) |
| CT alone | 10 (45) |
| RT alone | 2 (9) |
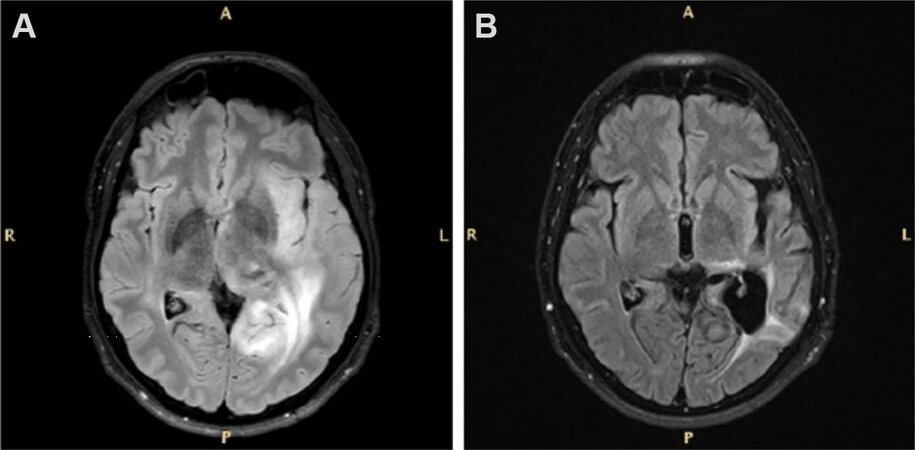
Patients included 22 males (67%) and 11 females (33%). ECOG PS was 0-1 in 21 patients (64%) and ≥ 2 in 12 patients (36%). Biopsy was performed in 22 cases (67%); at the histological analysis, 16 (73%) were astrocytomas and 6 (27%) were oligodendrogliomas. MGMT promoter methylation was detected in 14 patients (42%), and it was methylated in eight (57%). The IDH1 mutation status was studied in 16 out of 22 cases (73%), and it had mutated in 10 patients (63%). With regard to the type of treatment, 9 patients (27%) underwent concomitant RT plus TMZ, 22 patients (67%) only received TMZ, and 2 patients (6%) received RT alone; the choice of treatment was at the physician’s discretion, essentially based on ECOG PS and extent of disease. We then analyzed the best radiological response on the MRI, finding a complete response in 1 case (3%), partial response in 9 patients (27%) and stable disease in 15 patients (45%). Only eight patients (25%) had a progressive disease (Figure 1 shows a partial response in the case of diffuse astrocytoma).
Figure 1. FLAIR magnetic resonance images of a patient with diffuse astrocytoma before (A) and after (B) biopsy, and subsequent temozolomide treatment showing a partial response according to the RANO criteria for low-grade gliomas. FLAIR: Fluid-attenuated inversion recovery; RANO: Response Assessment in Neuro-Oncology.
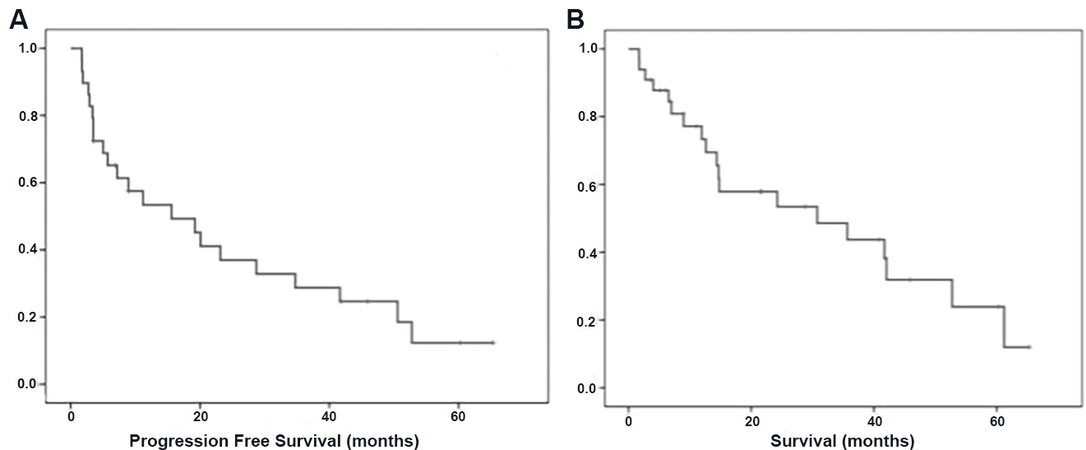
Finally, with regard to patient outcome, we found a PFS of 19.1 months and an OS of 30.7 months among all patients [Figure 2]; according to the ECOG PS, PFS was 34.6 months vs. 3.4 months for PS 0-1 and ≥ 2 (P < 0.0001), and OS was 42 months vs. 8.9 months (P < 0.0001), respectively [Table 2].
Figure 2. Median progression free survival of 19.1 months (A); and median overall survival of 30.7 months (B).
Univariate analysis for progression free survival and overall survival
| Variable | PFS | OS | ||||
| ECOG PS | P < 0.0001 | P < 0.0001 | ||||
| 0-1 | 34.6 | 8.7-60.6 | 42.0 | 27.7-56.3 | ||
| ≥ 2 | 3.4 | 0.69-6.2 | 8.9 | 5.5-12.39 | ||
| MGMT status | P = 0.05 | P = 0.009 | ||||
| Methylated | 41.6 | 8.35-74.9 | 52.7 | 40.4-54.6 | ||
| Unmethylated | 8.9 | 1.25-16.5 | 14.6 | 7.9-21.4 | ||
| IDH1 status | P = 0.006 | P = 0.2 | ||||
| Mutated | 52.7 | n.a | 57.7 | n.a | ||
| Wild-Type | 8.9 | 0.001-22.9 | 41.7 | n.a | ||
| First-line treatment | P = 0.2 | P = 0.7 | ||||
| TMZ + RT | 11.1 | 6.7-15.6 | 14.7 | 14.5-14.9 | ||
| RT or TMZ alone | 19.1 | 0.00-42.9 | 30.7 | 8.7-52.6 | ||
| Histological subtype | P = 0.7 | P = 0.8 | ||||
| Astrocytoma | 41.6 | 10.3-72.9 | 42.0 | 24.3-59.6 | ||
| Oligodendroglioma | 28.6 | 9.3-48.0 | 52.7 | n.a. | ||
| Best response | P < 0.0001 | P < 0.0001 | ||||
| PD | 2.7 | 1.3-4.1 | 4.0 | 2.69-14.6 | ||
| SD | 19.1 | 7.3-30.9 | 30.7 | 23.0-49.1 | ||
| PR + CR | 41.6 | 24.03-59.2 | 38.5 | 33.0-59.0 | ||
| Second-line treatment | P = 0.04 | |||||
| Yes | 30.7 | 6.8-54.5 | ||||
| No | 6.5 | 0.01-13.8 | ||||
With reference to the histological subtype, PFS between astrocytic and oligodendroglial tumors was 41.6 months vs. 28.6 months (P = 0.7), while OS was 42.0 months vs. 52.7 months (P = 0.8), respectively.
The presence of MGMT methylation was associated with longer PFS (41.6 months vs. 8.9 months, P = 0.05) and OS (52.7 months vs. 14.6 months, P = 0.009), while, in regard to IDH1 mutation, PFS was 52.7 months vs. 8.9 months (P = 0.006) and OS was 52.7 months vs. 41 months (P = 0.02).
It is worth noting that no significant difference was found in relation to the different types of treatment: for the concomitant scheme (RT + TMZ) vs. RT or TMZ alone, PFS was 11.1 months vs. 19.1 months (P = 0.2) and OS was 14.7 months vs. 30.7 months (P = 0.7), respectively.
Moreover, radiological response was correlated with survival; indeed, patients with partial or complete response had a longer OS than those with stable or progressive disease (38.5 months in CR/PR vs. 4.0 months in PD, P < 0.0001).
In our cases, 13 of 22 patients underwent second-line therapy after relapsing: of these, 1 patient received RT plus TMZ, while 10 patients received chemotherapy alone (fotemustine in four PT, PCV/PC in four PT, and TMZ in two PT) and 2 patients received RT alone; all of these patients had longer survival, with an OS of 30.7 months compared to 6.5 months for those who did not have second-line treatment (P = 0.04).
DISCUSSION
Gliomatosis cerebri is a rare entity, and it appears very difficult to define better management with multiple studies attempting to investigate it. This work illustrates the real-life management of this disease from a single institution, where the main goal was to deepen possible prognostic factors and treatment significance.
Based on a recent literature review by Carroll et al.[4], the median OS in GC is between 14.5 and 30 months, with an average of 21 months. Due to its heterogeneous nature, it is still difficult to define clear prognostic factors, although several studies have investigated them: a recent review reports that young age, low-grade histology, good PS, 1p19q codeletion, O6-methylguanin-DNA-methyltransferase (MGMT) promoter methylation, and IDH1-R132H-mutation seem to correlate with longer survival[5]. In fact, Desestret et al.[6] added that alpha-internexin (INA) expression, in particular when associated with IDH1 mutation, can also be a good prognostic factor.
Because of its infiltrative and diffuse spreading, the surgical approach is limited to biopsy or the minimal debulking of focal areas causing mass effect[7]. Radiotherapy (RT) has historically been used in GC, but its effectiveness is still unclear: multiple studies were conducted in this regard, but their results do not agree regarding survival advantage and dose/modalities of treatment; therefore, RT is often used in selected patients suitable to receive it, while weighing the expected benefits with potential neurocognitive side effects[8].
Conversely, chemotherapy (CT) seems to have an important significance in prolonging survival: historically, the regimen procarbazine-lomustine-vincristine (PCV) was the most used treatment, as also supported by the NOA-05 study[9]. However, this has been replaced, in most cases, by temozolomide, which has easier administration and better tolerability compared to PCV[10-12]; CT is usually prescribed as upfront therapy, but it may also be used after RT with a survival benefit[13] or during RT for GC with more aggressive aspects[10].
One limitation in our study is the lack of neurocognitive assessment; this should be an important factor to be analyzed when comparing efficacy results between chemotherapy alone and chemotherapy plus radiotherapy.
In our cases, it appears that only 67% of patients underwent surgery/biopsy; however, when possible, biopsy should be mandatory to analyze the molecular aspects of GC for choosing the best treatment; indeed, we showed that MGMT methylation can be an important prognostic factor in this tumor, and it is known that patients with MGMT methylation might benefit from temozolomide.
However, in our population, we reported a relatively small number of biopsies, and it could be due to the poor ECOG PS of the patients or the morphological characteristics of the tumor. A recent study analyzing 78 patients showed that a tumor involving the midline brain structure can correlate with a higher perioperative morbidity[14]. Hence, the non-surgical approach could also be an appropriate decision. Overall, our opinion is that biopsy should be mandatory for patients in good clinical condition to better define the tumor’s nature, prognosis, and the best treatment.
It could be an interesting perspective for future insights to investigate novel and more specific radiological techniques, such as positron emission tomography (PET-CT) or PET-MRI with amino acid tracers such as 18F-fluoro-ethyl-l-tyrosine (FET) and 11C-methyl-L-methionine (MET), which can probably better distinguish more aggressive tumor areas[15].
The reason a similar OS was obtained in patients with oligodendrogliomas and astrocytomas is not clear. Indeed, oligodendrogliomas should have a longer OS than astrocytoma. It is likely that our result could be due to the small number of patients analyzed in this study.
It is also interesting to note that no difference was found between the type of treatment at diagnosis; however, our results could be due to the small population analyzed or the retrospective design of this study. Indeed, the type of treatment was unbalanced among the patients: 67% of them received chemotherapy alone, as the choice of treatment was at the physician’s discretion, which could have influenced clinical outcomes. This is still a much debated topic, with multiple studies published with different results; in most of them, CT and RT seem to have the same results[16,17], so it is reasonable to recommend CT upfront with the aim of limiting neurotoxicity induced by RT[5]. As regards the chemotherapy regime, TMZ resulted safer than PCV with similar results in terms of PFS and OS[18]; however, PCV was the only schedule analyzed in a phase 3 randomized study in patients with anaplastic oligodendroglial tumors. Therefore, it could be correct to use temozolomide in patients with astrocytoma and PCV for an oligodendroglial tumor. However, it appears clear that it is absolutely important to identify predictive factors and focus the therapy on specific targets to obtain a better outcome with minimal toxicity. The recent randomized, phase 3 study (RTOG 9802) analyzing high-risk low-grade glioma patients treated with RT + PCV vs. RT alone demonstrated a longer OS for the combination regimen[19]. A subsequent genomic analysis[20] showed that IDH-mutant high-risk LGG, regardless of the 1p19q codeletion status, received benefits from the administration of chemotherapy; conversely, in our population, we showed no significant difference between combination or single treatment, although multivariate analysis was not performed due to the small number of patients. In conclusion, based on our results, GC patients with a good performance status, mutated IDH, and/or methylated MGMT should be treated with combination therapy, where possible. In the case of advanced disease where RT cannot be performed, CT should be evaluated in selected patients with molecular alterations and second-line therapy could be recommended for patients with a good clinical condition.
DECLARATIONS
Authors’ contributionsContributed to patient and data collection: Bellu L, Caccese M, Cerretti C, Berti F, Busato F, Parisi A, Padovan M, Zagonel V, Lombardi G
Wrote the paper: Bellu L, Lombardi G
Availability of data and materialsAll the data are recorded at Veneto Institute of Oncology. No public database exists.
Financial support and sponsorshipNot applicable.
Conflicts of interestAll authors declare that there are no conflicts of interest.
Ethical approval and consent to participateThe study was approved by the Veneto Institute of Oncology Ethics Commitee.
Consent for publicationAll the patients signed the informed consent form.
Copyright©The Author(s) 2021.
REFERENCES
1. Louis DN, Perry A, Reifenberger G, et al. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol (Berl) 2016;131:803-20.
2. Taillibert S, Chodkiewicz C, Laigle-Donadey F, Napolitano M, Cartalat-Carel S, Sanson M. Gliomatosis cerebri: a review of 296 cases from the ANOCEF Database and the Literature. J Neurooncol 2006;76:201-5.
3. van den Bent MJ, Wefel JS, Schiff D, et al. Response assessment in neuro-oncology (a report of the RANO group): assessment of outcome in trials of diffuse low-grade gliomas. Lancet Oncol 2011;12:583-93.
4. Carroll KT, Hirshman B, Ali MA, et al. Management and survival patterns of patients with gliomatosis cerebri: a SEER-based analysis. World Neurosurg 2017;103:186-93.
5. Wen PY, Macdonald DR, Reardon DA, et al. Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 2010;28:1963-72.
6. Desestret V, Ciccarino P, Ducray F, et al. Prognostic stratification of gliomatosis cerebri by IDH1R132H and INA expression. J Neurooncol 2011;105:219-24.
7. Greenfield JP, Castañeda Heredia A, George E, Kieran MW, Morales La Madrid A. Gliomatosis cerebri: a consensus summary report from the First International Gliomatosis cerebri Group Meeting, March 26-27, 2015, Paris, France. Pediatr Blood Cancer 2016;63:2072-7.
8. Chen S, Tanaka S, Giannini C, et al. Gliomatosis cerebri: clinical characteristics, management, and outcomes. J Neurooncol 2013;112:267-75.
9. Glas M, Bähr O, Felsberg J, et al. NOA-05 phase 2 trial of procarbazine and lomustine therapy in gliomatosis cerebri. Ann Neurol 2011;70:445-53.
10. Rudà R, Bertero L, Sanson M. Gliomatosis cerebri: a review. Curr Treat Options Neurol 2014;16:273.
11. Sanson M, Napolitano M, Cartalat-Carel S, Taillibert S. La gliomatose cérébrale. Rev Neurol (Paris) 2005;161:173-81.
12. Sanson M, Cartalat-Carel S, Taillibert S, et al. Initial chemotherapy in gliomatosis cerebri. Neurology 2004;63:270.
13. Kong DS, Kim ST, Lee JI, et al. Impact of adjuvant chemotherapy for gliomatosis cerebri. BMC Cancer 2010;10:424.
14. Shimony N, Shofty B, Ram Z, Grossman R. Perioperative risk assessment of patients with gliomatosis cerebri. World Neurosurg 2017;98:334-8.
15. Dunet V, Pomoni A, Hottinger A, Nicod-Lalonde M, Prior JO. Performance of 18F-FET versus 18F-FDG-PET for the diagnosis and grading of brain tumors: systematic review and meta-analysis. Neuro Oncol 2015;18:426-34.
16. Mattox AK, Lark AL, Adamson DC. Marked response of gliomatosis cerebri to temozolomide and whole brain radiotherapy. Clin Neurol Neurosurg 2012;114:299-306.
17. Baumert BG, Hegi ME, van den Bent MJ, et al. Temozolomide chemotherapy versus radiotherapy in high-risk low-grade glioma (EORTC 22033-26033): a randomised, open-label, phase 3 intergroup study. Lancet Oncol 2016;17:1521-32.
18. Wick W, Hartmann C, Engel C, et al. NOA-04 randomized phase III trial of sequential radiochemotherapy of anaplastic glioma with procarbazine, lomustine, and vincristine or temozolomide. J Clin Oncol 2009;27:5874-80.
19. Buckner JC, Shaw EG, Pugh SL, et al. Radiation plus procarbazine, CCNU, and vincristine in low-grade glioma. N Engl J Med 2016;374:1344-55.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Bellu L, Caccese M, Cerretti G, Berti F, Busato F, Parisi A, Padovan M, Zagonel V, Lombardi G. Gliomatosis cerebri: a monocentric real-life experience. J Cancer Metastasis Treat 2021;7:29. http://dx.doi.org/10.20517/2394-4722.2021.89
AMA Style
Bellu L, Caccese M, Cerretti G, Berti F, Busato F, Parisi A, Padovan M, Zagonel V, Lombardi G. Gliomatosis cerebri: a monocentric real-life experience. Journal of Cancer Metastasis and Treatment. 2021; 7: 29. http://dx.doi.org/10.20517/2394-4722.2021.89
Chicago/Turabian Style
Bellu, Luisa, Mario Caccese, Giulia Cerretti, Franco Berti, Fabio Busato, Alessandro Parisi, Marta Padovan, Vittorina Zagonel, Giuseppe Lombardi. 2021. "Gliomatosis cerebri: a monocentric real-life experience" Journal of Cancer Metastasis and Treatment. 7: 29. http://dx.doi.org/10.20517/2394-4722.2021.89
ACS Style
Bellu, L.; Caccese M.; Cerretti G.; Berti F.; Busato F.; Parisi A.; Padovan M.; Zagonel V.; Lombardi G. Gliomatosis cerebri: a monocentric real-life experience. J. Cancer. Metastasis. Treat. 2021, 7, 29. http://dx.doi.org/10.20517/2394-4722.2021.89
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 7 clicks
Cite This Article 7 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.