Tumors due to chronic exposure to benzene and biomarkers of exposure
Abstract
Due to occupational activities, many people are exposed to carcinogenic airborne pollutants, such as benzene. Furthermore, benzene is also present in cigarette smoke. The main cancers associated with benzene, toluene, ethylbenzene, and xylenes (BTEX) are lung cancer and malignancies of the blood. At initial stages, lung cancer is often asymptomatic, but progression into metastasis is a huge clinical concern. We performed a review on possible biomarkers for monitoring increased genome instability and downregulation of the immune system in connection with overexposure to BTEX in a subgroup of workers being exposed to BTEX at Brazilian gas stations. The workers are subject to routine blood exams every semester; however, early genomic assessment is not routine. We evaluated available tests to be applied, including measuring benzene-derived metabolites in urine, determination of oxidative and inflammatory markers in blood, immunophenotypical profiling, micronucleus test, comet (single-cell gel electrophoresis) assay, (molecular) cytogenetics, chromosomal microarray, epigenetic-changes oriented tests, determination of mitochondrial (mt)DNA copy numbers, and studies on miRNA-level. However, no consensus was reached about their application and which test combination might be suited best.
Keywords
INTRODUCTION
The International Agency for Research on Cancer classifies chemical, physical, and biological agents with respect to their potential to be risk factors for cancer into Groups 1-4, according to their carcinogenic potential in humans. Group 1 agents are definitely known to be carcinogenic; Group 2A are those likely to be carcinogenic; Group 2B comprises possibly carcinogenic agents; Group 3 are those not classifiable; and Group 4 are those probably being non-carcinogenic[1]. Many people around the world are exposed to complex mixtures of chemical substances that belong to Groups 1, 2A, and/or 2B due to occupational activities. In this concern, it is difficult to distinguish the isolated action of a single carcinogenic agent.
A typical example is gas station workers, who are exposed to a mixture of gasoline, especially benzene, toluene, ethylbenzene, and xylenes (BTEX), which contains benzene (Group 1), toluene (Group 2B), ethylbenzene (Group 2B), and xylenes (Group 3), all being polycyclic aromatic hydrocarbons (PAH)[2]. Benzene being volatile, stable, and colorless, is added up to 1% to gasoline; furthermore, diesel combustion releases benzene, among other toxic substances[3,4]. Chronic exposure to benzene causes leukemia [acute myelogenous leukemia (AML), acute lymphocytic leukemia (ALL), and chronic lymphocytic leukemia (CLL)], other blood malignancies [myelodysplastic syndrome (MDS), and multiple myeloma (MM)], and non-Hodgkin lymphoma[5,6]. Additionally, benzene most likely causes lung cancer[6]. Workers at gas stations and petroleum refineries, performing other industrial activities (shipping, automobile repair, shoe manufacture, etc.), fuel drivers, and traffic wardens are subject to PAH pollution[7]. Because of these industrial activities and the distribution of gasoline and diesel, benzene can be found as an airborne pollutant in industrial and city environments. Thus, beyond the above-mentioned workers, the environmental contamination can affect the health of the whole population near and far from benzene production industries, turning the benzene contamination into a real public health problem[8]. Finally, benzene is also present in cigarette smoke.
RATIONALE, AIM, AND METHOD
The etiology of cancer is complex. One of the possible causes is chronic exposure to genotoxic substances due to occupational activities, causing genomic instability and downregulation of the immune system.
The aim of this review is to summarize tests suitable to determine biomarkers that can be applied to follow the harmful effects of BTEX.
This is not a systematic review. The selection of papers was conducted according to the keywords “gas station worker” and “benzene exposure”, and the most relevant papers were selected.
SURVEILLANCE BIOMARKERS
As the increase in cancer risk due to benzene/BTEX is a real problem, health monitoring of the above-mentioned groups at risk is an unmet need. The identification of relevant biomarkers for people being exposed to these carcinogens due to occupational activities is necessary. In general, mechanisms by which carcinogens trigger cancer involve genotoxicity and/or immunosuppression. Benzene usually requires metabolic activation to become carcinogenic. Hereby, metabolic enzymes can transform benzene into potent toxic and carcinogenic electrophiles. These byproducts form addition products, commonly referred to as adducts, with cellular macromolecules including DNA, RNA, lipids, and proteins. Therefore, benzene and/or its metabolites may produce severe hazards to cells, including DNA damage, mutations, chromosomal instability, epigenetic alterations, altered DNA repair and metabolic activation, oxidative stress, and immunosuppression[9,10].
Regular blood tests are already in use for BTEX-exposed workers, as even low levels of benzene exposure may lead to white blood cells (lymphocyte and B cells) as well as granulocyte suppression[11]. Such abnormalities in the blood of gas station workers in Gondar town, Northwest Ethiopia[12] have been reported. Besides, monitoring of liver and renal functions in blood is also very important[13], as increased levels of hepatic markers [lactate dehydrogenase (LDH), bilirubin, and alanine aminotransferase (AST)] were found to increase in Brazilian gas station workers[14].
However, current blood tests do not allow the monitoring of DNA integrity under BTEX exposure. Therefore, it is important to develop better tests to monitor DNA damage and cancer risk in populations subjected to exposure to prevent the emergence of cancer after years of accumulating genetic alterations. Cancer progression occurring over a relatively long period of time is a multistep process that involves sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, reprogramming energy metabolism, evading immune suppression, inducing angiogenesis, and finally activating invasion and metastasis[15].
While the need for such tests is unquestionable, there is ongoing controversy about which is the best biomarker for monitoring BTEX-exposed workers. Most likely, more than one biomarker needs to be applied, and a standard setup for screening tests needs to be developed and approved. Here, we describe some current possibilities for general health, immune status, and genomic (in) stability related to BTEX exposure.
IMMUNOPHENOTYPICAL PROFILING
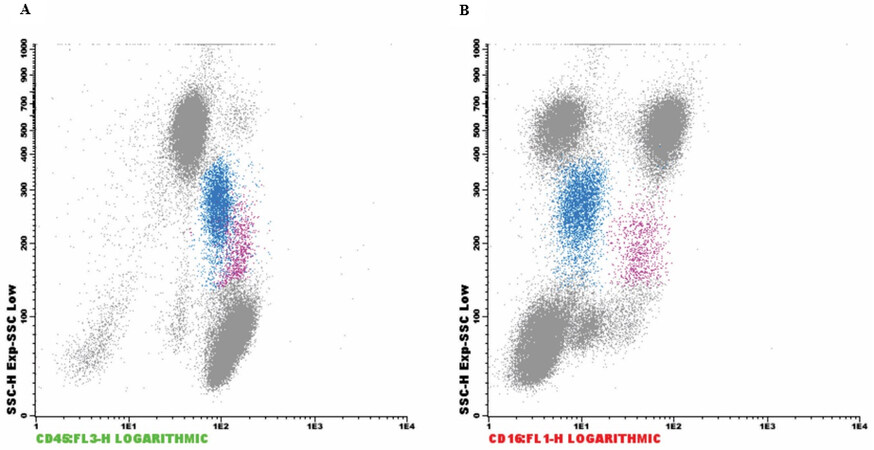
Immunophenotyping determines blood cell subpopulations by applying fluorescent-labeled monoclonal antibodies by flow cytometry. This would be an important complementary test besides the yet performed hemogram in the case of gas station workers, as highlighted by two female cases where natural killer (NK) cell count was found to be decreased, probably due to benzene exposure causing immunotoxicity[16]. Overall, NK cells are quite effective in controlling several types of tumors and microbial infections[17]. However, in another study, an increase of CD3+CD8+ T-lymphocytes was found with increased T memory helper cells (CD4+CD45RO) after BTEX exposure, but without alterations in NK cells[18]. In addition, other blood cell subpopulations could be affected, e.g., CD25+/CD4+ T lymphocytes were upregulated in BTEX-exposed refinery workers[19], while a decrease of CD80 and CD86 expression in monocytes or an increase of monocytes has also been reported[20,21]. Recently, our group detected activated monocytes in gas station workers in Rio de Janeiro [Figure 1].
MICRONUCLEUS TEST
The micronucleus (MN) test takes advantage of the fact that genomic damage can be visualized directly in lymphocyte nuclei of blood smears at a low cost; however, it requires the training of observers over the optical microscopy[7,14,22-24]. MNs are round, not larger than 0.5 µm in diameter, and, generally, only one is present per lymphocyte, besides the normal nucleus. MNs represent chromosomes that have been separated from the mitotic spindle during impaired mitosis and contain a high proportion of centromeric material along with heterochromatin[25]. MNs can also be assessed from exfoliated buccal mucosa cells[26-31] or cultures of peripheral blood lymphocytes[32].
An alternative in vitro assay is the cytokinesis-blocked micronucleus test. It is based on cultured lymphocytes being treated with cytochalasin B (Cyto-B), a cell-permeable mycotoxin disrupting the formation of actin polymers. Cells are harvested, fixed, and stained with Giemsa, and binucleated cells can be observed to determine the micronucleus frequencies[33].
COMET ASSAY (SINGLE-CELL GEL ELECTROPHORESIS)
In the comet assay, a single cell gel electrophoresis leads to the driving out of DNA from the nucleus. The microscopic image obtained is reminiscent of a comet. An enlarged comet tail is observed as a result of DNA fragmentation. The longer the tail, the greater the DNA fragmentation or DNA damage that has taken place[14,34]. The comet assay is a low-cost method with excellent sensitivity for the detection of DNA damage in gas station workers in Rio de Janeiro[14]. Comet assay is based on a few microliters of fresh blood, which is treated in a way that disrupts the cells on slides with agarose, following low-temperature alkaline electrophoresis. Finally, comets are classified, counted, and results are compared with lab internal standard values[35].
CHROMOSOMAL ABERRATIONS
Chromosomal aberrations (CAs) consist of breakage, rearrangements, and numerical variations (aneuploidy and polyploidy) that are visualized in the fixed chromosome preparations (metaphases). This cytogenetic analysis from peripheral blood lymphocytes for the determination of radiosensitivity or mutation load detection expressed as “frequency of CAs” is a long-standing approach[36]. A study conducted in the Czech Republic reinforced the association between increased levels of CAs and the incidence of cancer and mortality[37]. In BTEX-exposed workers, CAs in peripheral blood lymphocytes were already reported based on conventional/banding cytogenetics[22,38,39] or chromosome painting by fluorescence in situ hybridization (FISH)[40-46]. As for chromosome painting, whole chromosome probes are capable of labeling each pair of chromosomes with a different fluorescent color[47,48].
When chromosomes cannot be obtained in cell culture, an alternative is to study rearrangements in the interphase cell. Interphase FISH (I-FISH) requires the use of locus-specific probes and can only visualize small parts of the genome[49]. Moreover, new bioinformatics tools are available to evaluate cytogenomic data such as occurring CNV (copy number variation - see next section)[50] and can be applied in the future to detect instability in the genome of the BTEX-exposed workers.
Sister chromatic exchange (SCE) is another cytogenetics-based method that is suitable for the measurement of chromosomal rearrangements in benzene-exposed workers[19,32]. SCE reflects the interchange between DNA molecules at homologous loci within a replicating chromosome. The detection of SCEs in cytological preparations of metaphase chromosomes requires the incorporation of BrdU-dye[51,52]. A high number of SCEs (high frequency) may indicate chromosome instability.
CHROMOSOMAL MICROARRAY AND OTHER STUDIES TO ASSESS COPY NUMBER ALTERATIONS
Chromosomal analysis using an array platform can identify copy number variations (CNVs) in all chromosomes at a time. In addition, targeted assays may be used, as in the case of a study on Chinese benzene-exposed shoe factory workers without clinical symptoms. These workers were studied for CNVs in leukemia-related genes by chromosomal microarray (CMA) and real-time PCR analysis. The NOTCH1 and BSG mRNA levels were increased in benzene-exposed workers. These results suggest that CNVs and leukemia-related gene expression might play a role in leukemia development in such workers[53].
BENZENE-DERIVED METABOLITES IN URINE
Inhaled benzene is oxidized by CYP2E1 (belonging to the cytochrome P450 family) to a reactive intermediate benzene oxide, oxepine. This highly reactive species is further metabolized through three different pathways[54,55]:
(a) The predominant one leads to phenol via a non-enzymatic rearrangement. Phenol may be excreted in urine as sulfate or glucuronate derivatives (80%-90% of the absorbed dose) or may be oxidized via cytochrome P450 to 1,4- and 1,2-benzenediol. This may be further oxidized to p- and o-benzoquinone. The quinones may be back-reduced to the original diols via NADPH quinone oxidoreductase-1 (NQO1).
(b) The second pathway leads to benzene 1,2-dihydrodiol via epoxide hydrolase, which may be oxidized to trans,trans-muconic aldehyde and trans,trans-muconic acid (t,t-MA), which is excreted in urine (3%-18% of the adsorbed dose).
(c) The third and minor pathway leads to pre-phenyl mercapturic acid, after conjugation with glutathione via glutathione S-transferase (GST), and finally to S-phenylmercapturic acid (S-PMA), which is excreted in urine. Nonmetabolized benzene may also be excreted in urine (U-benzene) after passive diffusion from blood to urine through the kidney glomerular tuft.
To test for benzene exposure, t,t-MA and S-PMA are measured in urine samples by a very sensitive method[8,30,54,56]. Determination of urinary t,t-MA is carried out by pre-purification of urine with solid-phase extraction, followed by high-performance liquid chromatography and UV detection. Conversely, the determination of urinary S-PMA is based on an immunoassay technique. It should be noted that the main site of benzene metabolism is the liver, although the occurrence of benzene metabolism in the bone marrow should be the explanation for the frequently described myelotoxicity due to benzene exposure.
OXIDATIVE AND INFLAMMATORY MARKERS IN BLOOD
Inflammatory and oxidative stress biomarkers can be evaluated in blood samples of BTEX-exposed workers. Protein carbonyl (PCO) levels can be determined using a simple ELISA method, and the activity of GST is measured in 96-well microplates using a substrate (1-chloro-2,4-dinitrobenzene). While PCO was increased, the activity of GST was found to be reduced in Brazilian gas station workers[20]. GST is an important enzyme playing a role in detoxifying several carcinogens; thus, downregulation of GST activity can lead to genomic instability. In addition to GST, other metabolic and oxidative stress enzymes can be assessed, such as catalase (CAT), superoxide dismutase (SOD), thiol groups (THIOL), and malondialdehyde (MDA)[30].
From serum, inflammatory biomarkers can be assayed by ELISA:
EPIGENETICS
Epigenetic modifications (such as DNA-methylation) are partially influenced by environmental pollutants such as benzene. Epigenetic modification is important in carcinogenesis initiation and cancer progression. In humans, DNA methylation occurs at cytosine (C), which is transformed into 5-methylcytosine.
The target genes selected for methylation measurement are generally important for cell cycle control and DNA damage response and are related to detoxification metabolism. The cyclin-dependent kinase inhibitor 2A (CDKN2A) locus encodes two key tumor suppressor proteins (p14ARF and p16INK4A), which play a key role in the cell cycle control. Both p14ARF and p16INK4A were found hypermethylated in the mononuclear blood cells tested by MSP-PCR (Methylation-specific PCR)[57] in Brazilian gas station workers[58].
Repetitive DNA sequences can also be informative for epigenetic testing[30,59,61]. In a population of benzene-exposed workers, bisulfite-PCR pyrosequencing was used to quantify DNA methylation in repetitive DNA sequences (Line-1 and ALU), as well as in MAGE-1 and p15[59]. Exposure to benzene was associated with a reduction in LINE-1 and AluI methylation. In addition, hypermethylation in p15 and hypomethylation in MAGE-1 were detected.
MITOCHONDRIAL DNA COPY NUMBER
Mitochondrial DNA (mtDNA) is circular and present in multiple copies per mitochondrion. The mtDNA copy number is maintained within a constant range in normal cells, but in tumors as well as in age - related neurodegenerative disorders, the mtDNA copy number is highly variable[62]. mtDNA copy number variation has also been detected in workers exposed to hazardous compounds. Generally, the mtDNA copy number is determined by amplification of a region (e.g., ND1) from mtDNA based on qPCR (quantitative PCR) with a single copy gene (e.g., β-globin) as a reference. mtDNA was found to be increased in two studies linked to occupational exposure to benzene[31,63,64].
miRNA
MicroRNA (miRNA) are short non-coding RNAs that participate in the complex gene expression network[65]. Selected miRNAs play important roles in specific cancers, acting in the process of controlling signal transduction, cell development, differentiation, proliferation, apoptosis, and stress response. The expression of miR-221 was found to be elevated in lymphocytes of petrol station workers[66]. This is interesting because miR-221 has previously been found with elevated expression levels in leukemia patients, confirming miR-221 to be a potential biomarker for leukemogenesis.
In another work, the mechanism behind benzene-induced hematotoxicity was investigated, focusing on miRNA expression in benzene-poisoned Chinese workers. In blood lymphocyte-derived RNA, miR-222 was upregulated in the exposed workers and positively correlated with the induction of DNA strand breaks and MN formation. These results demonstrate a reduction of DNA repair capacity after benzene exposure, even in low doses[33]. Determination of miRNA expression can be performed by quantitative reverse transcriptase polymerase chain reaction (RT-qPCR)[33], while large-scale screening can be conducted by microarray assay platforms[33,66].
DISCUSSION
In a simple time scale, some of the mentioned biomarkers of exposure to BTEX can be considered more conventional ones (MN, comet test, molecular cytogenetics, measuring benzene-derived metabolites in urine, and measuring oxidative and inflammatory markers in blood), since they have been applied for decades. The more predictive biomarkers demand more sophisticated equipment or platforms (immunophenotypical profile, CMA, epigenetic evaluation, mtDNA copy number, and miRNA detection). The more predictive methods are generally more expensive and require specialized workers, and this is probably the main reason for not reaching a worldwide consensus method.
In Brazil, self-service at gas stations is forbidden, and consumers have to rely on gas station workers to fill their tanks. The newest official Brazilian legislation (https://www.in.gov.br/en/web/dou/-/portaria-n-427-de-7-de-outubro-de-2021-351525197), published on 7 October 2021, describes in detail all the necessary care in handling mixtures containing benzene including the worker’s personal protective equipment and the corresponding warnings and rights. However, while workers should have a hemogram done every semester, no other genetic-, cellular-, or metabolite-based monitoring tests are required or mentioned in the legislation. Here, the major concern is that early damages to lungs that might lead to cancer are not detectable by hemogram counts. Cancer is a multistep process that can take many years, while in the genome mutations, epigenetic modifications, and rearrangements are acquired, and the immune system is also impaired.
The detection of early lung cancer is desirable considering the higher chances of survival with lower costs of treatment. In our hospital, early lung cancer is detected by tomography or bronchoscopy. The application of new molecular-based methods would make the lung cancer diagnosis much more accurate and less invasive. With this aim, investigations are performed by liquid biopsy on cell-free DNA (cfDNA), all kinds of RNA (cfRNA), and volatile organic metabolites (VOMs). In the institutions where advanced molecular methods are available, the detection of cfDNA EGFR T790M mutation [responsible for resistance to gefitinib/erlotinib treatment in non-small cell lung cancer (NSCLC)] is already routine[67]. VOMs derived from differentiated cell metabolism are under investigation using biofluids (exhaled breath, pleural effusions, blood, urine, etc.) from lung cancer patients and can be captured by solid-phase microextraction combined with gas chromatography-mass spectrometry (SPME-GC/MS)[68].
Even better than early lung cancer detection is monitoring the increasing genome instability by biomarkers (genetic, immunological, and metabolite), taking action to prevent worker overexposure to BTEX, and consequently protecting them from developing cancer. This review is important because it contributes to the discussion of the Brazilian scientists and lawmakers (possibly extended to other countries) on what would be the best protocol to monitor the genomic stability and the health of the immune system of BTEX-exposed workers, as well as what actions should be taken to prevent cancer in these workers.
CONCLUSION
Many promising biomarkers for BTEX exposure are available to monitor genetic and immunological alterations leading to leukemia and lung cancer. Thus far, there is no universal consensus on one or more gold standard biomarkers to be assessed. Overall, it is recommended to apply more than one biomarker for greater accuracy. Without a consensus, the biomarker choice (genetic, immunological, or metabolite) depends on the tools available in each laboratory and/or affordable for the country. Thus, the available biomarkers should be analyzed along with clinical examinations, regular blood exams such as the hemogram, and biochemical markers.
DECLARATIONS
Authors’ contributionsMade substantial contributions to the conception and design of the work: Alves G
Revised the manuscript critically and added important intellectual content: Alves G, Nunes R, Melo K, Ornellas MHF
Availability of data and materialsNot available.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022
REFERENCES
1. International Agency for Research on Cancer (IARC). Monographs on the evaluation of carcinogenic risks to humans - benzene volume 120. 2018. Available from: https://publications.iarc.fr/Book-And-Report-Series/Iarc-Monographs-On-The-Identification-Of-Carcinogenic-Hazards-To-Humans/Benzene-2018 [Last accessed on 17 May 2022].
2. International Agency for Research on Cancer (IARC). Agents classified by the IARC monographs, volumes 1–129. Available from: https://monographs.iarc.who.int/agents-classified-by-the-iarc/ [Last accessed on 17 May 2022].
3. The National Institute for Occupational Safety and Health (NIOSH). Carcinogenic effects of exposure to diesel exhaust. 1988. Available from: https://www.cdc.gov/niosh/docs/88-116/default.html [Last accessed on 17 May 2022].
4. International Agency for Research on Cancer (IARC). Diesel and gasoline engine exhausts and some nitroarenes. 2014. Available from: https://publications.iarc.fr/129 [Last accessed on 17 May 2022].
5. Houot MT, Homère J, Goulard H, Garras L, Delabre L, Pilorget C. Lifetime occupational exposure proportion estimation methods: a sensitivity analysis in the general population. Int Arch Occup Environ Health 2021;94:1537-47.
6. International Agency for Research on Cancer (IARC). 2020. Available from: https://monographs.iarc.who.int/wp-content/uploads/2019/07/Classifications_by_cancer_site.pdf [Last accessed on 17 May 2022].
7. Angelini S, Maffei F, Bermejo JL, et al. Environmental exposure to benzene, micronucleus formation and polymorphisms in DNA-repair genes: a pilot study. Mutat Res 2012;743:99-104.
8. Silva CB, Mota CL, Almeida YR, et al. Environmental exposure to benzene: evaluation of urinary S-PMA and polymorphism (CYP2E1-1293G>C and NQO1 609C>T) in Campos Elíseos residents, Duque de Caxias, Rio de Janeiro State, Brazil. Cad Saude Publica 2019;35:e00198618.
9. Smith MT, Guyton KZ, Gibbons CF, et al. Key Characteristics of carcinogens as a basis for organizing data on mechanisms of carcinogenesis. Environ Health Perspect 2016;124:713-21.
11. Lan Q, Zhang L, Li G, et al. Hematotoxicity in workers exposed to low levels of benzene. Science 2004;306:1774-6.
12. Getu S, Shiferaw E, Melku M. Assessment of hematological parameters of petrol filling workers at petrol stations in Gondar town, Northwest Ethiopia: a comparative cross-sectional study. Environ Health Prev Med 2020;25:44.
13. Abou-ElWafa HS, Albadry AA, El-Gilany AH, Bazeed FB. Some biochemical and hematological parameters among petrol station attendants: a comparative study. Biomed Res Int 2015;2015:418724.
14. Melo K, Santiago F, Lucena SBG et al. Aplastic anemia, clinical implications and DNA damage in workers with occupational exposure to aromatic hydrocarbons in Rio de Janeiro. Res Results Biomed 2020;6:308-17.
16. Santiago F, Lima S, Pinheiro T, et al. Benzene poisoning, clinical and blood abnormalities in two Brazilian female gas station attendants: two case reports. BMC Res Notes 2017;10:52.
17. Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S. Functions of natural killer cells. Nat Immunol 2008;9:503-10.
18. Brandão MM, Rêgo MA, Pugliese L, et al. Phenotype analysis of lymphocytes of workers with chronic benzene poisoning. Immunol Lett 2005;101:65-70.
19. Biró A, Pállinger É, Major J, et al. Lymphocyte phenotype analysis and chromosome aberration frequency of workers occupationally exposed to styrene, benzene, polycyclic aromatic hydrocarbons or mixed solvents. Immunol Lett 2002;81:133-40.
20. Moro AM, Sauer E, Brucker N, et al. Evaluation of immunological, inflammatory, and oxidative stress biomarkers in gasoline station attendants. BMC Pharmacol Toxicol 2019;20:75.
21. Silvestre RT, Delmonico L, Bravo M, et al. Health survey and assessment of the polymorphisms BRCA1/P871L, BRCA1/Q356R, and BRCA2/N372H in female gas station workers in Rio de Janeiro. Environ Mol Mutagen 2017;58:730-4.
22. Zhang L, Eastmond DA, Smith MT. The nature of chromosomal aberrations detected in humans exposed to benzene. Crit Rev Toxicol 2002;32:1-42.
23. Chandirasekar R, Kumar BL, Sasikala K, et al. Assessment of genotoxic and molecular mechanisms of cancer risk in smoking and smokeless tobacco users. Mutat Res Genet Toxicol Environ Mutagen 2014;767:21-7.
24. Lovreglio P, Maffei F, Carrieri M, et al. Evaluation of chromosome aberration and micronucleus frequencies in blood lymphocytes of workers exposed to low concentrations of benzene. Mutat Res Genet Toxicol Environ Mutagen 2014;770:55-60.
25. Mohandas N. Structure and composition of the erythrocyte. In: Kaushansky K, Lichtman MA, Prchal JT, Levi MM, Oliver WP, Burns LJ, Caligiuri, editors. Williams Hematology. 9th edition. New York: Mcgraw Hill, 2016; pp. 467-68.
26. Celik A, Cavaş T, Ergene-Gözükara S. Cytogenetic biomonitoring in petrol station attendants: micronucleus test in exfoliated buccal cells. Mutagenesis 2003;18:417-21.
27. Singaraju M, Singaraju S, Parwani R, Wanjari S. Cytogenetic biomonitoring in petrol station attendants: A micronucleus study. J Cytol 2012;29:1-5.
28. Shaikh A, Barot D, Chandel D. Genotoxic Effects of exposure to gasoline fumes on petrol pump workers. Int J Occup Environ Med 2018;9:79-87.
29. Filho APR, Silveira MAD, do Nascimento CB, d'Arce LPG. Integrative study of cell damage and cancer risk in gas station attendants. Int J Environ Health Res 2018;28:1-7.
30. Costa-Amaral IC, Carvalho LVB, Santos MVC, et al. Environmental assessment and evaluation of oxidative stress and genotoxicity biomarkers related to chronic occupational exposure to benzene. Int J Environ Res Public Health 2019;16:2240.
31. Gaikwad AS, Mahmood R, Beerappa R, Karunamoorthy P, Venugopal D. Mitochondrial DNA copy number and cytogenetic damage among fuel filling station attendants. Environ Mol Mutagen 2020;61:820-9.
32. Mrdjanović J, Šolajić S, Dimitrijević S, Đan I, Nikolić I, Jurišić V. Assessment of micronuclei and sister chromatid exchange frequency in the petroleum industry workers in province of Vojvodina, Republic of Serbia. Food Chem Toxicol 2014;69:63-8.
33. Wang TS, Tian W, Fang Y, et al. Changes in miR-222 expression, DNA repair capacity, and MDM2-p53 axis in association with low-dose benzene genotoxicity and hematotoxicity. Sci Total Environ 2021;765:142740.
34. Ostling O, Johanson K. Microelectrophoretic study of radiation-induced DNA damages in individual mammalian cells. Biochem Biophysci Res Commun 1984;123:291-8.
35. Collins AR. The Comet Assay for DNA Damage and repair: principles, applications, and limitations. Mol Biotechnol 2004;26:249-61.
36. Rossner P, Boffetta P, Ceppi M, et al. Chromosomal aberrations in lymphocytes of healthy subjects and risk of cancer. Environ Health Perspect 2005;113:517-20.
37. Smerhovsky Z, Landa K, Rössner P, et al. Risk of cancer in an occupationally exposed cohort with increased level of chromosomal aberrations. Environ Health Perspect 2001;109:41-5.
38. Gonçalves RO, de Almeida Melo N, Rêgo MA. Association between occupational exposure to benzene and chromosomal alterations in lymphocytes of Brazilian petrochemical workers removed from exposure. Environ Monit Assess 2016;188:334.
39. Villalba-Campos M, Chuaire-Noack L, Sánchez-Corredor MC, Rondón-Lagos M. High chromosomal instability in workers occupationally exposed to solvents and paint removers. Mol Cytogenet 2016;9:46.
40. Verdorfer I, Neubauer S, Letzel S, et al. Chromosome painting for cytogenetic monitoring of occupationally exposed and non-exposed groups of human individuals. Mut Res/Gene Toxicol Environ Mutagene 2001;491:97-109.
41. Zhang L, Lan Q, Guo W, et al. Use of OctoChrome fluorescence in situ hybridization to detect specific aneuploidy among all 24 chromosomes in benzene-exposed workers. Chem Biol Interact 2005;153-154:117-22.
42. Holeckova B, Piesova E, Sivikova K, Dianovsky J. FISH detection of chromosome 1 aberration in human interphase and metaphase lymphocytes after exposure to benzene. Ann Agric Environ Med 2008;15:99-103.
43. Zhang L, Lan Q, Guo W, et al. Chromosome-wide aneuploidy study (CWAS) in workers exposed to an established leukemogen, benzene. Carcinogenesis 2011;32:605-12.
44. Ji Z, Zhang L. Chromosomics: detection of numerical and structural alterations in all 24 human chromosomes simultaneously using a novel OctoChrome FISH assay. J Vis Exp ;2012:3619.
45. Santiago F, Alves G, Otero UB, et al. Monitoring of gas station attendants exposure to benzene, toluene, xylene (BTX) using three-color chromosome painting. Mol Cytogenet 2014;7:15.
46. Santiago F, Silvestre RT, Otero UB, et al. The association of three DNA repair genes polymorphisms on the frequency of chromosomal alterations detected by fluorescence in situ hybridization. Int Arch Occup Environ Health 2021;94:1567-77.
47. Liehr T, Claussen U. FISH-technology. lab manual. FISH on chromosome preparations of peripheral blood, Berlin: Springer, 2002. pp. 73–81.
48. Liehr T, Kosyakova N. Part IV Multicolor-FISH-Probe sets (MFISH) and immunostaining - multiplex FISH and spectral karyotyping. In: Liehr T, editor, Fluorescence in situ Hybridization (FISH), 2th edition, Berlin: Springer, 2017, pp. 233-39.
49. Vorsanova SG, Yurov YB, Iourov IY. Human interphase chromosomes: a review of available molecular cytogenetic technologies. Mol Cytogenet 2010;3:1.
50. Iourov IY, Vorsanova SG, Yurov YB. In silico molecular cytogenetics: a bioinformatic approach to prioritization of candidate genes and copy number variations for basic and clinical genome research. Mol Cytogenet 2014;7:98.
51. Wolff S, Perry P. Differential Giemsa staining of sister chromatids and the study of chromatid exchanges without autoradiography. Chromosoma 1974;48:341-53.
52. Goto K, Maeda S, Kano Y, Sugiyama T. Factors involved in differential Giemsa-staining of sister chromatids. Chromosoma 1978;66:351-9.
53. Li K, Jing Y, Yang C, et al. Increased leukemia-associated gene expression in benzene-exposed workers. Sci Rep 2014;4:5369.
54. Fustinoni S, Consonni D, Campo L, et al. Monitoring low benzene exposure: comparative evaluation of urinary biomarkers, influence of cigarette smoking, and genetic polymorphisms. Cancer Epidemiol Biomarkers Prev 2005;14:2237-44.
55. Bruckner JV, Anaud SS, Warren AD. Toxic effects of solvents and vapors, chapter 24. In: Casarett and Doul´s Toxicology, the basic science of poisons, 7th edition. New York: McGraw-Hill, 2008; pp. 1008-1009.
56. Zhang L, Ye FL, Chen T, Mei Y, Song SZ. Trans, trans-muconic acid as a biomarker of occupational exposure to high-level benzene in China. J Occup Environ Med 2011;53:1194-8.
57. Herman JG, Graff JR, Myöhänen S, Nelkin BD, Baylin SB. Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc Natl Acad Sci USA 1996;93:9821-6.
58. Silvestre RT, Bravo M, Santiago F, et al. Hypermethylation in gene promoters are induced by chronic exposure to benzene, toluene, ethylbenzene and xylenes. Pak J Biol Sci 2020;23:518-25.
59. Bollati V, Baccarelli A, Hou L, et al. Changes in DNA methylation patterns in subjects exposed to low-dose benzene. Cancer Res 2007;67:876-80.
60. Peluso M, Bollati V, Munnia A, et al. DNA methylation differences in exposed workers and nearby residents of the Ma Ta Phut industrial estate, Rayong, Thailand. Int J Epidemiol 2012;41:1753-60; discussion 1761.
61. Byun HM, Motta V, Panni T, et al. Evolutionary age of repetitive element subfamilies and sensitivity of DNA methylation to airborne pollutants. Part Fibre Toxicol 2013;10:28.
62. Filograna R, Mennuni M, Alsina D, Larsson NG. Mitochondrial DNA copy number in human disease: the more the better? FEBS Lett 2021;595:976-1002.
63. Shen M, Zhang L, Bonner MR, et al. Association between mitochondrial DNA copy number, blood cell counts, and occupational benzene exposure. Environ Mol Mutagen 2008;49:453-7.
64. Carugno M, Pesatori AC, Dioni L, et al. Increased mitochondrial DNA copy number in occupations associated with low-dose benzene exposure. Environ Health Perspect 2012;120:210-5.
65. Khan S, Jha A, Panda AC, Dixit A. Cancer-Associated circRNA-miRNA-mRNA Regulatory Networks: A Meta-Analysis. Front Mol Biosci 2021;8:671309.
66. Hu D, Peng X, Liu Y, et al. Overexpression of miR-221 in peripheral blood lymphocytes in petrol station attendants: A population based cross-sectional study in southern China. Chemosphere 2016;149:8-13.
67. Rosell R, Karachaliou N. Implications of blood-based T790M genotyping and beyond in epidermal growth factor Receptor-Mutant Non-Small-Cell Lung Cancer. J Clin Oncol 2016;34:3361-2.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Alves G, Nunes RA, Melo K, Ornellas MHF. Tumors due to chronic exposure to benzene and biomarkers of exposure. J Cancer Metastasis Treat 2022;8:20. http://dx.doi.org/10.20517/2394-4722.2022.02
AMA Style
Alves G, Nunes RA, Melo K, Ornellas MHF. Tumors due to chronic exposure to benzene and biomarkers of exposure. Journal of Cancer Metastasis and Treatment. 2022; 8(5): 20. http://dx.doi.org/10.20517/2394-4722.2022.02
Chicago/Turabian Style
Alves, Gilda, Rodolfo Acatauassú Nunes, Karina Melo, Maria Helena Faria Ornellas. 2022. "Tumors due to chronic exposure to benzene and biomarkers of exposure" Journal of Cancer Metastasis and Treatment. 8, no.5: 20. http://dx.doi.org/10.20517/2394-4722.2022.02
ACS Style
Alves, G.; Nunes RA.; Melo K.; Ornellas MHF. Tumors due to chronic exposure to benzene and biomarkers of exposure. J. Cancer. Metastasis. Treat. 2022, 8, 20. http://dx.doi.org/10.20517/2394-4722.2022.02
About This Article
Copyright
Data & Comments
Data

 Cite This Article 7 clicks
Cite This Article 7 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.